
History of the molecule
Encyclopedia
Chemistry
Chemistry is the science of matter, especially its chemical reactions, but also its composition, structure and properties. Chemistry is concerned with atoms and their interactions with other atoms, and particularly with the properties of chemical bonds....
, the history of molecular theory traces the origins of the concept or idea of the existence of strong chemical bonds
Covalent bond
A covalent bond is a form of chemical bonding that is characterized by the sharing of pairs of electrons between atoms. The stable balance of attractive and repulsive forces between atoms when they share electrons is known as covalent bonding....
between two or more atom
Atom
The atom is a basic unit of matter that consists of a dense central nucleus surrounded by a cloud of negatively charged electrons. The atomic nucleus contains a mix of positively charged protons and electrically neutral neutrons...
s.
The modern concept of molecules can be traced back towards pre-scientific Greek philosophers such as Leucippus
Leucippus
Leucippus or Leukippos was one of the earliest Greeks to develop the theory of atomism — the idea that everything is composed entirely of various imperishable, indivisible elements called atoms — which was elaborated in greater detail by his pupil and successor, Democritus...
who argued that all the universe is composed of atoms and voids
Atomic theory
In chemistry and physics, atomic theory is a theory of the nature of matter, which states that matter is composed of discrete units called atoms, as opposed to the obsolete notion that matter could be divided into any arbitrarily small quantity...
. Circa 450 BC Empedocles
Empedocles
Empedocles was a Greek pre-Socratic philosopher and a citizen of Agrigentum, a Greek city in Sicily. Empedocles' philosophy is best known for being the originator of the cosmogenic theory of the four Classical elements...
imagined fundamental elements (fire
Fire (classical element)
Fire has been an important part of all cultures and religions from pre-history to modern day and was vital to the development of civilization. It has been regarded in many different contexts throughout history, but especially as a metaphysical constant of the world.-Greek and Roman tradition:Fire...
(
 ), earth
), earthEarth (classical element)
Earth, home and origin of humanity, has often been worshipped in its own right with its own unique spiritual tradition.-European tradition:Earth is one of the four classical elements in ancient Greek philosophy and science. It was commonly associated with qualities of heaviness, matter and the...
(
 ), air
), airAir (classical element)
Air is often seen as a universal power or pure substance. Its supposed fundamental importance to life can be seen in words such as aspire, inspire, perspire and spirit, all derived from the Latin spirare.-Greek and Roman tradition:...
(
 ), and water
), and waterWater (classical element)
Water is one of the elements in ancient Greek philosophy, in the Asian Indian system Panchamahabhuta, and in the Chinese cosmological and physiological system Wu Xing...
(
 )) and "forces" of attraction and repulsion allowing the elements to interact. Prior to this, Heraclitus
)) and "forces" of attraction and repulsion allowing the elements to interact. Prior to this, HeraclitusHeraclitus
Heraclitus of Ephesus was a pre-Socratic Greek philosopher, a native of the Greek city Ephesus, Ionia, on the coast of Asia Minor. He was of distinguished parentage. Little is known about his early life and education, but he regarded himself as self-taught and a pioneer of wisdom...
had claimed that fire or change was fundamental to our existence, created through the combination of opposite properties. In the Timaeus
Timaeus (dialogue)
Timaeus is one of Plato's dialogues, mostly in the form of a long monologue given by the title character, written circa 360 BC. The work puts forward speculation on the nature of the physical world and human beings. It is followed by the dialogue Critias.Speakers of the dialogue are Socrates,...
, Plato
Plato
Plato , was a Classical Greek philosopher, mathematician, student of Socrates, writer of philosophical dialogues, and founder of the Academy in Athens, the first institution of higher learning in the Western world. Along with his mentor, Socrates, and his student, Aristotle, Plato helped to lay the...
, following Pythagoras
Pythagoras
Pythagoras of Samos was an Ionian Greek philosopher, mathematician, and founder of the religious movement called Pythagoreanism. Most of the information about Pythagoras was written down centuries after he lived, so very little reliable information is known about him...
, considered mathematical entities such as number, point, line and triangle as the fundamental building blocks or elements of this ephemeral world, and considered the four elements of fire, air, water and earth as states of substances through which the true mathematical principles or elements would pass. A fifth element, the incorruptible quintessence aether
Aether (classical element)
According to ancient and medieval science aether , also spelled æther or ether, is the material that fills the region of the universe above the terrestrial sphere.-Mythological origins:...
, was considered to be the fundamental building block of the heavenly bodies. The viewpoint of Leucippus and Empedocles, along with the aether, was accepted by Aristotle
Aristotle
Aristotle was a Greek philosopher and polymath, a student of Plato and teacher of Alexander the Great. His writings cover many subjects, including physics, metaphysics, poetry, theater, music, logic, rhetoric, linguistics, politics, government, ethics, biology, and zoology...
and passed to medieval and renaissance Europe. A modern conceptualization of molecules began to develop in the 19th century along with experimental evidence for pure chemical elements and how individual atoms of different chemical substances such as hydrogen and oxygen can combine to form chemically stable molecules such as water molecules.
17th century
The earliest views on the shapes and connectivity of atoms was that proposed by LeucippusLeucippus
Leucippus or Leukippos was one of the earliest Greeks to develop the theory of atomism — the idea that everything is composed entirely of various imperishable, indivisible elements called atoms — which was elaborated in greater detail by his pupil and successor, Democritus...
, Democritus
Democritus
Democritus was an Ancient Greek philosopher born in Abdera, Thrace, Greece. He was an influential pre-Socratic philosopher and pupil of Leucippus, who formulated an atomic theory for the cosmos....
, and Epicurus
Epicurus
Epicurus was an ancient Greek philosopher and the founder of the school of philosophy called Epicureanism.Only a few fragments and letters remain of Epicurus's 300 written works...
who reasoned that the solidness of the material corresponded to the shape of the atoms involved. Thus, iron atoms are solid and strong with hooks that lock them into a solid; water atoms are smooth and slippery; salt atoms, because of their taste, are sharp and pointed; and air atoms are light and whirling, pervading all other materials. It was Democritus that was the main proponent of this view. Using analogies from our sense experiences, he gave a picture or an image of an atom in which atoms were distinguished from each other by their shape, their size, and the arrangement of their parts. Moreover, connections were explained by material links in which single atoms were supplied with attachments: some with hooks and eyes others with balls and sockets.
With the rise of Christianity and the decline of the Roman Empire, the atomic theory was abandoned for nearly two millennia in favor of the various four element theories and later alchemical theories. The 17th century, however, saw a resurgence in the atomic theory primarily through the works of Descartes
René Descartes
René Descartes ; was a French philosopher and writer who spent most of his adult life in the Dutch Republic. He has been dubbed the 'Father of Modern Philosophy', and much subsequent Western philosophy is a response to his writings, which are studied closely to this day...
, Gassendi
Pierre Gassendi
Pierre Gassendi was a French philosopher, priest, scientist, astronomer, and mathematician. With a church position in south-east France, he also spent much time in Paris, where he was a leader of a group of free-thinking intellectuals. He was also an active observational scientist, publishing the...
, and Newton
Isaac Newton
Sir Isaac Newton PRS was an English physicist, mathematician, astronomer, natural philosopher, alchemist, and theologian, who has been "considered by many to be the greatest and most influential scientist who ever lived."...
. Using earlier Greek atomic theories to explain how the tiniest particles of matter bonded together, Descartes visualized that atoms were held together by microscopic hooks and barbs. He held that two atoms combined when the hook of one got caught in the eye of the other (see diagram):
By the mid 1770s, it was generally believed that any theory involving particles endowed with physical hooks was considered “Cartesian chemistry”. Similar to Descartes, Gassendi, who had recently written a book on the life of Epicurus, reasoned that to account for the size and shape of atoms moving in a void could account for the properties of matter. Heat was due to small, round atoms; cold, to pyramidal atoms with sharp points, which accounted for the pricking sensation of severe cold; and solids were held together by interlacing hooks.
Newton, though he acknowledged the various atom attachment theories in vogue at the time, i.e. “hooked atoms”, “glued atoms” (bodies at rest), and the “stick together by conspiring motions” theory, rather believed, as famously stated in "Query 31" of his 1704 Opticks
Opticks
Opticks is a book written by English physicist Isaac Newton that was released to the public in 1704. It is about optics and the refraction of light, and is considered one of the great works of science in history...
, that particles attract one another by some force, which “in immediate contact is extremely strong, at small distances performs the chemical operations, and reaches not far from particles with any sensible effect.”
In a more concrete manner, however, the concept of aggregates or units of bonded atoms, i.e. "molecules", traces its origins to Robert Boyle
Robert Boyle
Robert Boyle FRS was a 17th century natural philosopher, chemist, physicist, and inventor, also noted for his writings in theology. He has been variously described as English, Irish, or Anglo-Irish, his father having come to Ireland from England during the time of the English plantations of...
's 1661 hypothesis, in his famous treatise The Sceptical Chymist, that matter is composed of clusters of particles and that chemical change results from the rearrangement of the clusters. Boyle argued that matter's basic elements consisted of various sorts and sizes of particles, called "corpuscle
Corpuscle
Corpuscle may refer to:*a small free floating biological cell, especially a blood cell, but not a fat cell*a nerve ending such as Meissner's corpuscle or a Pacinian corpuscle*any member of Corpus Christi College, Cambridge or Corpus Christi College, Oxford...
s", which were capable of arranging themselves into groups.
In 1680, using the corpuscular theory
Corpuscular theory
In optics, corpuscular theory of light, set forward by Sir Isaac Newton, states that light is made up of small discrete particles called "corpuscles" which travel in a straight line with a finite velocity and possess kinetic energy....
as a basis, French chemist Nicolas Lemery
Nicolas Lemery
Nicolas Lémery , French chemist, was born at Rouen. He was one of the first to develop theories on acid-base chemistry....
stipulated that the acidity of any substance consisted in its pointed particles, while alkalis were endowed with pores of various sizes. A molecule, according to this view, consisted of corpuscles united through a geometric locking of points and pores.
18th century
Chemical affinity
In chemical physics and physical chemistry, chemical affinity is the electronic property by which dissimilar chemical species are capable of forming chemical compounds...
". For example, in 1718, building on Boyle’s conception of combinations of clusters, the French chemist Étienne François Geoffroy
Étienne François Geoffroy
Étienne François Geoffroy , French physician and chemist, best known for his 1718 affinity tables. He first contemplated a career as an apothecary, but then decided to practice medicine. He is sometimes known as Geoffroy the Elder.-Biography:He was born in Paris...
developed theories of chemical affinity
Chemical affinity
In chemical physics and physical chemistry, chemical affinity is the electronic property by which dissimilar chemical species are capable of forming chemical compounds...
to explain combinations of particles, reasoning that a certain alchemical “force” draws certain alchemical components together. Geoffroy's name is best known in connection with his tables of "affinities
Chemical affinity
In chemical physics and physical chemistry, chemical affinity is the electronic property by which dissimilar chemical species are capable of forming chemical compounds...
" (tables des rapports), which he presented to the French Academy in 1718 and 1720.
These were lists, prepared by collating observations on the actions of substances one upon another, showing the varying degrees of affinity exhibited by analogous bodies for different reagent
Reagent
A reagent is a "substance or compound that is added to a system in order to bring about a chemical reaction, or added to see if a reaction occurs." Although the terms reactant and reagent are often used interchangeably, a reactant is less specifically a "substance that is consumed in the course of...
s. These tables retained their vogue for the rest of the century, until displaced by the profounder conceptions introduced by CL Berthollet
Claude Louis Berthollet
Claude Louis Berthollet was a Savoyard-French chemist who became vice president of the French Senate in 1804.-Biography:...
.
In 1738, Swiss physicist and mathematician Daniel Bernoulli
Daniel Bernoulli
Daniel Bernoulli was a Dutch-Swiss mathematician and was one of the many prominent mathematicians in the Bernoulli family. He is particularly remembered for his applications of mathematics to mechanics, especially fluid mechanics, and for his pioneering work in probability and statistics...
published Hydrodynamica, which laid the basis for the kinetic theory
Kinetic theory
The kinetic theory of gases describes a gas as a large number of small particles , all of which are in constant, random motion. The rapidly moving particles constantly collide with each other and with the walls of the container...
of gases. In this work, Bernoulli positioned the argument, still used to this day, that gas
Gas
Gas is one of the three classical states of matter . Near absolute zero, a substance exists as a solid. As heat is added to this substance it melts into a liquid at its melting point , boils into a gas at its boiling point, and if heated high enough would enter a plasma state in which the electrons...
es consist of great numbers of molecules moving in all directions, that their impact on a surface causes the gas pressure
Pressure
Pressure is the force per unit area applied in a direction perpendicular to the surface of an object. Gauge pressure is the pressure relative to the local atmospheric or ambient pressure.- Definition :...
that we feel, and that what we experience as heat
Heat
In physics and thermodynamics, heat is energy transferred from one body, region, or thermodynamic system to another due to thermal contact or thermal radiation when the systems are at different temperatures. It is often described as one of the fundamental processes of energy transfer between...
is simply the kinetic energy of their motion. The theory was not immediately accepted, in part because conservation of energy
Conservation of energy
The nineteenth century law of conservation of energy is a law of physics. It states that the total amount of energy in an isolated system remains constant over time. The total energy is said to be conserved over time...
had not yet been established, and it was not obvious to physicists how the collisions between molecules could be perfectly elastic.
In 1789, William Higgins
William Higgins (chemist)
William Higgins , an Irish chemist, was one of the early proponents of atomic theory. Known mainly for his speculative ideas on chemical combination, William Higgins is popular for the insights his life offers into the emergence of chemistry as a career during the British industrial revolution...
published views on what he called combinations of "ultimate" particles, which foreshadowed the concept of valency bonds. If, for example, according to Higgins, the force between the ultimate particle of oxygen and the ultimate particle of nitrogen were 6, then the strength of the force would be divided accordingly, and similarly for the other combinations of ultimate particles:

19th century
Similar to these views, in 1803 John DaltonJohn Dalton
John Dalton FRS was an English chemist, meteorologist and physicist. He is best known for his pioneering work in the development of modern atomic theory, and his research into colour blindness .-Early life:John Dalton was born into a Quaker family at Eaglesfield, near Cockermouth, Cumberland,...
took the atomic weight of hydrogen, the lightest element, as unity, and determined, for example, that the ratio for nitrous anhydride was 2 to 3 which gives the formula N2O3. Interestingly, Dalton incorrectly imagined that atoms “hooked” together to form molecules. Later, in 1808, Dalton published his famous diagram of combined "atoms":
In Amedeo Avogadro
Amedeo Avogadro
Lorenzo Romano Amedeo Carlo Avogadro di Quaregna e di Cerreto, Count of Quaregna and Cerreto was an Italian savant. He is most noted for his contributions to molecular theory, including what is known as Avogadro's law...
's famous 1811 paper "Essay on Determining the Relative Masses of the Elementary Molecules of Bodies", he essentially states, i.e. according to Partington
J. R. Partington
James Riddick Partington MBE was a British chemist and historian of chemistry.- Life and work :Partington was born in Bolton, Lancashire and was educated at the University of Manchester, where he obtained First Class Honours...
's A Short History of Chemistry, that:
Note that this quote is not a literal translation. Avogadro uses the name "molecule" for both atoms and molecules. Specifically, he uses the name "elementary molecule" when referring to atoms and to complicate the matter also speaks of "compound molecules" and "composite molecules".
During his stay in Vercelli, Avogadro wrote a concise note (memoria) in which he declared the hypothesis of what we now call Avogadro's law
Avogadro's law
Avogadro's law is a gas law named after Amedeo Avogadro who, in 1811, hypothesized that two given samples of an ideal gas, at the same temperature, pressure and volume, contain the same number of molecules...
: equal volumes of gases, at the same temperature and pressure, contain the same number of molecules. This law implies that the relationship occurring between the weights of same volumes of different gases, at the same temperature and pressure, corresponds to the relationship between respective molecular weights. Hence, relative molecular masses could now be calculated from the masses of gas samples.
Avogadro developed this hypothesis in order to reconcile Joseph Louis Gay-Lussac
Joseph Louis Gay-Lussac
- External links :* from the American Chemical Society* from the Encyclopædia Britannica, 10th Edition * , Paris...
's 1808 law on volumes and combining gases with Dalton's 1803 atomic theory
Atomic theory
In chemistry and physics, atomic theory is a theory of the nature of matter, which states that matter is composed of discrete units called atoms, as opposed to the obsolete notion that matter could be divided into any arbitrarily small quantity...
. The greatest difficulty Avogadro had to resolve was the huge confusion at that time regarding atoms and molecules—one of the most important contributions of Avogadro's work was clearly distinguishing one from the other, admitting that simple particles too could be composed of molecules, and that these are composed of atoms. Dalton, by contrast, did not consider this possibility. Curiously, Avogadro considers only molecules containing even numbers of atoms; he does not say why odd numbers are left out.
In 1826, building on the work of Avogadro, the French chemist Jean-Baptiste Dumas
Jean-Baptiste Dumas
Jean Baptiste André Dumas was a French chemist, best known for his works on organic analysis and synthesis, as well as the determination of atomic weights and molecular weights by measuring vapor densities...
states:
In coordination with these concepts, in 1833 the French chemist Marc Antoine Auguste Gaudin
Marc Antoine Auguste Gaudin
Marc Antoine Auguste Gaudin was an French chemist.He was a pioneer in photography and contributed to the Avogadro gas theory by proposing that some elements form diatomic or polyatomic gas.-References:...
presented a clear account of Avogadro's hypothesis, regarding atomic weights, by making use of “volume diagrams”, which clearly show both semi-correct molecular geometries, such as a linear water molecule, and correct molecular formulas, such as H2O:
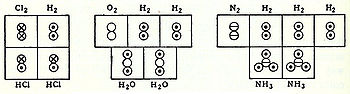
In 1856, Scottish chemist Archibald Couper began research on the bromination of benzene at the laboratory of Charles Wurtz in Paris. One month after Kekulé's second paper appeared, Couper's independent and largely identical theory of molecular structure was published. He offered a very concrete idea of molecular structure, proposing that atoms joined to each other like modern-day Tinkertoys in specific three-dimensional structures. Couper was the first to use lines between atoms, in conjunction with the older method of using brackets, to represent bonds, and also postulated straight chains of atoms as the structures of some molecules, ring-shaped molecules of others, such as in tartaric acid
Tartaric acid
Tartaric acid is a white crystalline diprotic organic acid. It occurs naturally in many plants, particularly grapes, bananas, and tamarinds; is commonly combined with baking soda to function as a leavening agent in recipes, and is one of the main acids found in wine. It is added to other foods to...
and cyanuric acid
Cyanuric acid
Cyanuric acid or 1,3,5-triazine-2,4,6-triol is a chemical compound with the formula 3. Like many industrially useful chemicals, this triazine has many synonyms. This white, odorless solid finds use as a precursor or a component of bleaches, disinfectants, and herbicides...
In later publications, Couper’s bonds were represented using straight dotted lines (although it is not known if this is the typesetter’s preference) such as with alcohol
Alcohol
In chemistry, an alcohol is an organic compound in which the hydroxy functional group is bound to a carbon atom. In particular, this carbon center should be saturated, having single bonds to three other atoms....
and oxalic acid
Oxalic acid
Oxalic acid is an organic compound with the formula H2C2O4. This colourless solid is a dicarboxylic acid. In terms of acid strength, it is about 3,000 times stronger than acetic acid. Oxalic acid is a reducing agent and its conjugate base, known as oxalate , is a chelating agent for metal cations...
below:
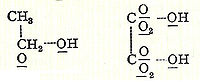
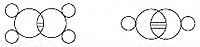
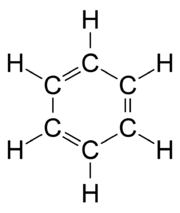
Kathleen Lonsdale
Dame Kathleen Lonsdale, DBE FRS was a crystallographer, who established the structure of benzene by X-ray diffraction methods in 1929, and hexachlorobenzene by Fourier spectral methods in 1931...
. Benzene presents a special problem in that, to account for all the bonds, there must be alternating double
Double bond
A double bond in chemistry is a chemical bond between two chemical elements involving four bonding electrons instead of the usual two. The most common double bond, that between two carbon atoms, can be found in alkenes. Many types of double bonds between two different elements exist, for example in...
carbon bonds:
August Wilhelm von Hofmann
August Wilhelm von Hofmann was a German chemist.-Biography:Hofmann was born at Gießen, Grand Duchy of Hesse. Not intending originally to devote himself to physical science, he first took up the study of law and philology at Göttingen. But he then turned to chemistry, and studied under Justus von...
was the first to make stick-and-ball molecular models, which he used in lecture at the Royal Institution of Great Britain, such as methane shown below:
The basis of this model followed the earlier 1855 suggestion by his colleague William Odling
William Odling
William Odling, FRS was an English chemist who contributed to the development of the periodic table....
that carbon
Carbon
Carbon is the chemical element with symbol C and atomic number 6. As a member of group 14 on the periodic table, it is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds...
is tetravalent. Hofmann's color scheme, to note, is still used to this day: nitrogen
Nitrogen
Nitrogen is a chemical element that has the symbol N, atomic number of 7 and atomic mass 14.00674 u. Elemental nitrogen is a colorless, odorless, tasteless, and mostly inert diatomic gas at standard conditions, constituting 78.08% by volume of Earth's atmosphere...
= blue, oxygen
Oxygen
Oxygen is the element with atomic number 8 and represented by the symbol O. Its name derives from the Greek roots ὀξύς and -γενής , because at the time of naming, it was mistakenly thought that all acids required oxygen in their composition...
= red, chlorine
Chlorine
Chlorine is the chemical element with atomic number 17 and symbol Cl. It is the second lightest halogen, found in the periodic table in group 17. The element forms diatomic molecules under standard conditions, called dichlorine...
= green, sulfur
Sulfur
Sulfur or sulphur is the chemical element with atomic number 16. In the periodic table it is represented by the symbol S. It is an abundant, multivalent non-metal. Under normal conditions, sulfur atoms form cyclic octatomic molecules with chemical formula S8. Elemental sulfur is a bright yellow...
= yellow, hydrogen
Hydrogen
Hydrogen is the chemical element with atomic number 1. It is represented by the symbol H. With an average atomic weight of , hydrogen is the lightest and most abundant chemical element, constituting roughly 75% of the Universe's chemical elemental mass. Stars in the main sequence are mainly...
= white. The deficiencies in Hofmann's model were essentially geometric: carbon
Carbon
Carbon is the chemical element with symbol C and atomic number 6. As a member of group 14 on the periodic table, it is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds...
bonding was shown as planar, rather than tetrahedral, and the atoms were out of proportion, e.g. carbon was smaller in size than the hydrogen.
In 1864, Scottish organic chemist Alexander Crum Brown
Alexander Crum Brown
Alexander Crum Brown FRSE FRS was a Scottish organic chemist.-Biography:Born in Edinburgh, the half-brother of the physician and essayist John Brown, he studied for five years at the Royal High School, succeeded by one year at Mill Hill School in London...
began to draw pictures of molecules, in which he enclosed the symbols for atoms in circles, and used broken lines to connect the atoms together in a way that satisfied each atom's valence.
The year 1873, by many accounts, was a seminal point in the history of the development of the concept of the "molecule". In this year, the renowned Scottish physicist James Clerk Maxwell
James Clerk Maxwell
James Clerk Maxwell of Glenlair was a Scottish physicist and mathematician. His most prominent achievement was formulating classical electromagnetic theory. This united all previously unrelated observations, experiments and equations of electricity, magnetism and optics into a consistent theory...
published his famous thirteen page article 'Molecules' in the September issue of Nature. In the opening section to this article, Maxwell clearly states:
After speaking about the atomic theory
Atomic theory
In chemistry and physics, atomic theory is a theory of the nature of matter, which states that matter is composed of discrete units called atoms, as opposed to the obsolete notion that matter could be divided into any arbitrarily small quantity...
of Democritus
Democritus
Democritus was an Ancient Greek philosopher born in Abdera, Thrace, Greece. He was an influential pre-Socratic philosopher and pupil of Leucippus, who formulated an atomic theory for the cosmos....
, Maxwell goes on to tell us that the word 'molecule' is a modern word. He states, "it does not occur in Johnson's Dictionary. The ideas it embodies are those belonging to modern chemistry." We are told that an 'atom' is a material point, invested and surrounded by 'potential forces' and that when 'flying molecules' strike against a solid body in constant succession it causes what is called pressure
Pressure
Pressure is the force per unit area applied in a direction perpendicular to the surface of an object. Gauge pressure is the pressure relative to the local atmospheric or ambient pressure.- Definition :...
of air and other gases. At this point, however, Maxwell notes that no one has ever seen or handled a molecule.
In 1874, Jacobus Henricus van 't Hoff
Jacobus Henricus van 't Hoff
Jacobus Henricus van 't Hoff, Jr. was a Dutch physical and organic chemist and the first winner of the Nobel Prize in chemistry. He is best known for his discoveries in chemical kinetics, chemical equilibrium, osmotic pressure, and stereochemistry...
and Joseph Achille Le Bel
Joseph Achille Le Bel
Joseph Achille Le Bel was a French chemist. He is best known for his work in stereochemistry. Le Bel was educated at the École Polytechnique in Paris. In 1874 he announced his theory outlining the relationship between molecular structure and optical activity...
independently proposed that the phenomenon of optical activity could be explained by assuming that the chemical bonds between carbon atoms and their neighbors were directed towards the corners of a regular tetrahedron. This led to a better understanding of the three-dimensional nature of molecules.
Emil Fischer
Emil Fischer
Emil Fischer may refer to:* Emil Fischer , German dramatic basso* Franz Joseph Emil Fischer , German chemist, worked with oil and coal* Hermann Emil Fischer , German Nobel laureate in chemistry...
developed the Fischer projection
Fischer projection
The Fischer projection, devised by Hermann Emil Fischer in 1891, is a two-dimensional representation of a three-dimensional organic molecule by projection. Fischer projections were originally proposed for the depiction of carbohydrates and used by chemists, particularly in organic chemistry and...
technique for viewing 3-D molecules on a 2-D sheet of paper:

Ludwig Boltzmann
Ludwig Eduard Boltzmann was an Austrian physicist famous for his founding contributions in the fields of statistical mechanics and statistical thermodynamics...
, in his Lectures on Gas Theory, used the theory of valence
Valence (chemistry)
In chemistry, valence, also known as valency or valence number, is a measure of the number of bonds formed by an atom of a given element. "Valence" can be defined as the number of valence bonds...
to explain the phenomenon of gas phase molecular dissociation, and in doing so drew one of the first rudimentary yet detailed atomic orbital overlap drawings. Noting first the known fact that molecular iodine
Iodine
Iodine is a chemical element with the symbol I and atomic number 53. The name is pronounced , , or . The name is from the , meaning violet or purple, due to the color of elemental iodine vapor....
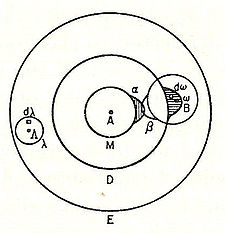
vapor dissociates into atoms at higher temperatures, Boltzmann states that we must explain the existence of molecules composed of two atoms, the “double atom” as Boltzmann calls it, by an attractive force acting between the two atoms. Boltzmann states that this chemical attraction, owing to certain facts of chemical valence, must be associated with a relatively small region on the surface of the atom called the sensitive region.
Boltzmann states that this "sensitive region" will lie on the surface of the atom, or may partially lie inside the atom, and will firmly be connected to it. Specifically, he states “only when two atoms are situated so that their sensitive regions are in contact, or partly overlap, will there be a chemical attraction between them. We then say that they are chemically bound to each other.” This picture is detailed below, showing the α-sensitive region of atom-A overlapping with the β-sensitive region of atom-B:
20th century
In the early 20th century, the American chemist Gilbert N. LewisGilbert N. Lewis
Gilbert Newton Lewis was an American physical chemist known for the discovery of the covalent bond , his purification of heavy water, his reformulation of chemical thermodynamics in a mathematically rigorous manner accessible to ordinary chemists, his theory of Lewis acids and...
began to use dots in lecture, while teaching undergraduates at Harvard, to represent the electrons around atoms. His students favored these drawings, which stimulated him in this direction. From these lectures, Lewis noted that elements with a certain number of electrons seemed to have a special stability. This phenomenon was pointed out by the German chemist Richard Abegg
Richard Abegg
Richard Wilhelm Heinrich Abegg was a German chemist and pioneer of valence theory. He proposed that the difference of the maximum positive and negative valence of an element tends to be eight. This has come to be called Abegg's rule...
in 1904, to which Lewis referred to as "Abegg's law of valence" (now generally known as Abegg's rule
Abegg's rule
In chemistry, Abegg’s rule states that the difference between the maximum positive and negative valence of an element is frequently eight. The rule used a historic meaning of valence which resembles the modern concept of oxidation state in which an atom is an electron donor or receiver...
). To Lewis it appeared that once a core of eight electrons has formed around a nucleus, the layer is filled, and a new layer is started. Lewis also noted that various ion
Ion
An ion is an atom or molecule in which the total number of electrons is not equal to the total number of protons, giving it a net positive or negative electrical charge. The name was given by physicist Michael Faraday for the substances that allow a current to pass between electrodes in a...
s with eight electrons also seemed to have a special stability. On these views, he proposed the rule of eight or octet rule
Octet rule
The octet rule is a chemical rule of thumb that states that atoms of low The octet rule is a chemical rule of thumb that states that atoms of low The octet rule is a chemical rule of thumb that states that atoms of low (The octet rule is a chemical rule of thumb that states that atoms of low (...
: Ions or atoms with a filled layer of eight electrons have a special stability.
Moreover, noting that a cube has eight corners Lewis envisioned an atom as having eight sides available for electrons, like the corner of a cube. Subsequently, in 1902 he devised a conception in which cubic atoms can bond on their sides to form cubic-structured molecules.
In other words, electron-pair bonds are formed when two atoms share an edge, as in structure C below. This results in the sharing of two electrons. Similarly, charged ionic-bonds are formed by the transfer of an electron from one cube to another, without sharing an edge A. An intermediate state B where only one corner is shared was also postulated by Lewis.
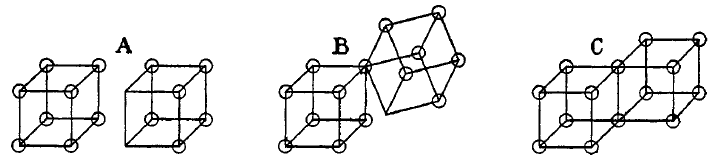
Double bond
A double bond in chemistry is a chemical bond between two chemical elements involving four bonding electrons instead of the usual two. The most common double bond, that between two carbon atoms, can be found in alkenes. Many types of double bonds between two different elements exist, for example in...
s are formed by sharing a face between two cubic atoms. This results in the sharing of four electrons.
In 1913, while working as the chair of the department of chemistry at the University of California, Berkeley
University of California, Berkeley
The University of California, Berkeley , is a teaching and research university established in 1868 and located in Berkeley, California, USA...
, Lewis read a preliminary outline of paper by an English graduate student, Alfred Lauck Parson
Alfred Lauck Parson
Alfred Lauck Parson was a British chemist and physicist, whose "magneton theory" of the atom contributed to the history of chemistry.-Biography:...
, who was visiting Berkeley for a year. In this paper, Parson suggested that the electron
Electron
The electron is a subatomic particle with a negative elementary electric charge. It has no known components or substructure; in other words, it is generally thought to be an elementary particle. An electron has a mass that is approximately 1/1836 that of the proton...
is not merely an electric charge but is also a small magnet (or "magneton
Toroidal ring model
The toroidal ring model, known originally as the Parson magneton or magnetic electron, is also known as the plasmoid ring, vortex ring, or helicon ring...
" as he called it) and furthermore that a chemical bond
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electromagnetic force attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction...
results from two electrons being shared between two atoms. This, according to Lewis, meant that bonding occurred when two electrons formed a shared edge between two complete cubes.
On these views, in his famous 1916 article The Atom and the Molecule, Lewis introduced the “Lewis structure” to represent atoms and molecules, where dots represent electron
Electron
The electron is a subatomic particle with a negative elementary electric charge. It has no known components or substructure; in other words, it is generally thought to be an elementary particle. An electron has a mass that is approximately 1/1836 that of the proton...
s and lines represent covalent bond
Covalent bond
A covalent bond is a form of chemical bonding that is characterized by the sharing of pairs of electrons between atoms. The stable balance of attractive and repulsive forces between atoms when they share electrons is known as covalent bonding....
s. In this article, he developed the concept of the electron-pair bond
Covalent bond
A covalent bond is a form of chemical bonding that is characterized by the sharing of pairs of electrons between atoms. The stable balance of attractive and repulsive forces between atoms when they share electrons is known as covalent bonding....
, in which two atoms may share one to six electrons, thus forming the single electron bond, a single bond, a double bond
Double bond
A double bond in chemistry is a chemical bond between two chemical elements involving four bonding electrons instead of the usual two. The most common double bond, that between two carbon atoms, can be found in alkenes. Many types of double bonds between two different elements exist, for example in...
, or a triple bond
Triple bond
A triple bond in chemistry is a chemical bond between two chemical elements involving six bonding electrons instead of the usual two in a covalent single bond. The most common triple bond, that between two carbon atoms, can be found in alkynes. Other functional groups containing a triple bond are...
.
In Lewis' own words:
Moreover, he proposed that an atom tended to form an ion by gaining or losing the number of electrons needed to complete a cube. Thus, Lewis structures show each atom in the structure of the molecule using its chemical symbol. Lines are drawn between atoms that are bonded to one another; occasionally, pairs of dots are used instead of lines. Excess electrons that form lone pairs are represented as pair of dots, and are placed next to the atoms on which they reside:
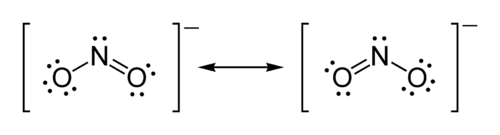
The following year, in 1917, an unknown American undergraduate chemical engineer named Linus Pauling
Linus Pauling
Linus Carl Pauling was an American chemist, biochemist, peace activist, author, and educator. He was one of the most influential chemists in history and ranks among the most important scientists of the 20th century...
was learning the Dalton hook-and-eye bonding method at the Oregon Agricultural College, which was the vogue description of bonds between atoms at the time. Each atom had a certain number of hooks that allowed it to attach to other atoms, and a certain number of eyes that allowed other atoms to attach to it. A chemical bond resulted when a hook and eye connected. Pauling, however, wasn't satisfied with this archaic method and looked to the newly-emerging field of quantum physics for a new method.
In 1927, the physicists Fritz London
Fritz London
Fritz Wolfgang London was a German theoretical physicist. His fundamental contributions to the theories of chemical bonding and of intermolecular forces are today considered classic and are discussed in standard textbooks of physical chemistry.With his brother Heinz, he made a significant...
and Walter Heitler
Walter Heitler
Walter Heinrich Heitler was a German physicist who made contributions to quantum electrodynamics and quantum field theory...
applied the new quantum mechanics to the deal with the saturable, nondynamic forces of attraction and repulsion, i.e., exchange forces, of the hydrogen molecule. Their valence bond treatment of this problem, in their joint paper, was a landmark in that it brought chemistry under quantum mechanics. Their work was an influence on Pauling, who had just received his doctorate and visited Heitler and London in Zürich on a Guggenheim Fellowship
Guggenheim Fellowship
Guggenheim Fellowships are American grants that have been awarded annually since 1925 by the John Simon Guggenheim Memorial Foundation to those "who have demonstrated exceptional capacity for productive scholarship or exceptional creative ability in the arts." Each year, the foundation makes...
.
Subsequently, in 1931, building on the work of Heitler and London and on theories found in Lewis' famous article, Pauling published his ground-breaking article "The Nature of the Chemical Bond" (see: manuscript) in which he used quantum mechanics
Quantum mechanics
Quantum mechanics, also known as quantum physics or quantum theory, is a branch of physics providing a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter. It departs from classical mechanics primarily at the atomic and subatomic...
to calculate properties and structures of molecules, such as angles between bonds and rotation about bonds. On these concepts, Pauling developed hybridization theory to account for bonds in molecules such as CH4, in which four sp³ hybridised orbitals are overlapped by hydrogen
Hydrogen
Hydrogen is the chemical element with atomic number 1. It is represented by the symbol H. With an average atomic weight of , hydrogen is the lightest and most abundant chemical element, constituting roughly 75% of the Universe's chemical elemental mass. Stars in the main sequence are mainly...
's 1s orbital, yielding four sigma (σ) bonds
Sigma bond
In chemistry, sigma bonds are the strongest type of covalent chemical bond. They are formed by head-on overlapping between atomic orbitals. Sigma bonding is most clearly defined for diatomic molecules using the language and tools of symmetry groups. In this formal approach, a σ-bond is...
. The four bonds are of the same length and strength, which yields a molecular structure as shown below:
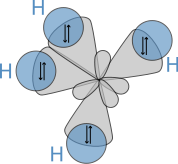
Nobel Prize in Chemistry
The Nobel Prize in Chemistry is awarded annually by the Royal Swedish Academy of Sciences to scientists in the various fields of chemistry. It is one of the five Nobel Prizes established by the will of Alfred Nobel in 1895, awarded for outstanding contributions in chemistry, physics, literature,...
. Notably he has been the only person to ever win two unshared Nobel prize
Nobel Prize
The Nobel Prizes are annual international awards bestowed by Scandinavian committees in recognition of cultural and scientific advances. The will of the Swedish chemist Alfred Nobel, the inventor of dynamite, established the prizes in 1895...
s, winning the Nobel Peace Prize
Nobel Peace Prize
The Nobel Peace Prize is one of the five Nobel Prizes bequeathed by the Swedish industrialist and inventor Alfred Nobel.-Background:According to Nobel's will, the Peace Prize shall be awarded to the person who...
in 1963.
In 1926, French physicist Jean Perrin received the Nobel Prize in physics for proving, conclusively, the existence of molecules. He did this by calculating Avogadro's number
Avogadro's number
In chemistry and physics, the Avogadro constant is defined as the ratio of the number of constituent particles N in a sample to the amount of substance n through the relationship NA = N/n. Thus, it is the proportionality factor that relates the molar mass of an entity, i.e...
using three different methods, all involving liquid phase systems. First, he used a gamboge
Gamboge
Gamboge is a partially transparent dark mustard yellow pigment.Other forms and spellings are: cambodia, cambogium, camboge, cambugium, gambaugium, gambogia, gambozia, gamboidea, gambogium, gumbouge, gambouge, gamboge, gambooge, gambugia...
soap-like emulsion, second by doing experimental work on Brownian motion
Brownian motion
Brownian motion or pedesis is the presumably random drifting of particles suspended in a fluid or the mathematical model used to describe such random movements, which is often called a particle theory.The mathematical model of Brownian motion has several real-world applications...
, and third by confirming Einstein’s theory of particle rotation in the liquid phase.
In 1937, chemist K.L. Wolf introduced the concept of supermolecules (Übermoleküle) to describe hydrogen bonding in acetic acid
Acetic acid
Acetic acid is an organic compound with the chemical formula CH3CO2H . It is a colourless liquid that when undiluted is also called glacial acetic acid. Acetic acid is the main component of vinegar , and has a distinctive sour taste and pungent smell...
dimers. This would eventually lead to the area of supermolecular chemistry, which is the study of non-covalent bonding.
In 1951, physicist Erwin Wilhelm Müller
Erwin Wilhelm Müller
Erwin Wilhelm Müller was a German physicist who invented the Field Emission Electron Microscope , the Field Ion Microscope , and the Atom-Probe Field Ion Microscope...
invents the field ion microscope
Field ion microscope
Field ion microscopy is an analytical technique used in materials science. The field ion microscope is a type of microscope that can be used to image the arrangement of atoms at the surface of a sharp metal tip....
and is the first to see atom
Atom
The atom is a basic unit of matter that consists of a dense central nucleus surrounded by a cloud of negatively charged electrons. The atomic nucleus contains a mix of positively charged protons and electrically neutral neutrons...
s, e.g. bonded atomic arrangements at the tip of a metal point.
In 1999, researchers from the University
University
A university is an institution of higher education and research, which grants academic degrees in a variety of subjects. A university is an organisation that provides both undergraduate education and postgraduate education...
of Vienna
Vienna
Vienna is the capital and largest city of the Republic of Austria and one of the nine states of Austria. Vienna is Austria's primary city, with a population of about 1.723 million , and is by far the largest city in Austria, as well as its cultural, economic, and political centre...
reported results from experiments on wave-particle duality for C60 molecules. The data published by Zeilinger et al. were consistent with de Broglie wave interference for C60 molecules. This experiment was noted for extending the applicability of wave–particle duality by about one order of magnitude in the macroscopic direction.
In 2009, researchers from IBM
IBM
International Business Machines Corporation or IBM is an American multinational technology and consulting corporation headquartered in Armonk, New York, United States. IBM manufactures and sells computer hardware and software, and it offers infrastructure, hosting and consulting services in areas...
managed to take the first picture of a real molecule. Using an Atomic Force Microscope
Atomic force microscope
Atomic force microscopy or scanning force microscopy is a very high-resolution type of scanning probe microscopy, with demonstrated resolution on the order of fractions of a nanometer, more than 1000 times better than the optical diffraction limit...
every single atom and bond of a pentacene
Pentacene
Pentacene is a polycyclic aromatic hydrocarbon consisting of five linearly-fused benzene rings. This highly conjugated compound is an organic semiconductor. The compound generates excitons upon absorption of ultra-violet or visible light; this makes it very sensitive to oxidation...
molecule could be imaged.
See also
- History of chemistryHistory of chemistryBy 1000 BC, ancient civilizations used technologies that would eventually form the basis of the various branches of chemistry. Examples include extracting metals from ores, making pottery and glazes, fermenting beer and wine, making pigments for cosmetics and painting, extracting chemicals from...
- History of quantum mechanicsHistory of quantum mechanicsThe history of quantum mechanics, as it interlaces with the history of quantum chemistry, began essentially with a number of different scientific discoveries: the 1838 discovery of cathode rays by Michael Faraday; the 1859-1860 winter statement of the black body radiation problem by Gustav...
- History of thermodynamicsHistory of thermodynamicsThe history of thermodynamics is a fundamental strand in the history of physics, the history of chemistry, and the history of science in general...
- History of molecular biologyHistory of molecular biologyThe history of molecular biology begins in the 1930s with the convergence of various, previously distinct biological disciplines: biochemistry, genetics, microbiology, and virology...
- Kinetic theoryKinetic theoryThe kinetic theory of gases describes a gas as a large number of small particles , all of which are in constant, random motion. The rapidly moving particles constantly collide with each other and with the walls of the container...
- Atomic theoryAtomic theoryIn chemistry and physics, atomic theory is a theory of the nature of matter, which states that matter is composed of discrete units called atoms, as opposed to the obsolete notion that matter could be divided into any arbitrarily small quantity...
External links
- Geometric Structures of Molecules - Middlebury College
- Atoms and Molecules - McMaster University
- 3D Molecule Viewer - The Wileys Family
- Molecule of the Month - School of Chemistry, University of Bristol
Types
- Antibody Molecule - The National Health Museum
- 15 Types of Molecules - IUPAC Definitions
Definitions
- Molecule Definition - Frostburg State UniversityFrostburg State UniversityFrostburg State University is a four-year university located on a campus in Frostburg, Maryland, in Western Maryland, and is part of the University System of Maryland. FSU is accredited by the Middle States Commission on Higher Education.-History:...
(Department of Chemistry) - Definition of Molecule - IUPAC
Articles
- Molecules Used to Make Nano-sized Containers - TRN Newswire
- Molecular Computer Processors - HP Labs

