
Coupling reaction
Encyclopedia
A coupling reaction in organic chemistry
is a catch-all term for a variety of reactions where two hydrocarbon
fragments are coupled with the aid of a metal catalyst. In one important reaction type a main group organometallic compound of the type RM (R = organic fragment, M = main group centre) reacts with an organic halide of the type R'X with formation of a new carbon-carbon bond
in the product R-R' Contributions to coupling reactions by Ei-ichi Negishi
and Akira Suzuki
were recognized with the 2010 Nobel Prize in Chemistry, which was shared with Richard F. Heck
.
Broadly speaking, two types of coupling reactions are recognized:
usually begins with oxidative addition
of one organic halide to the catalyst. Subsequently, the second partner undergoes transmetallation, which places both coupling partners on the same metal centre. The final step is reductive elimination of the two coupling fragments to regenerate the catalyst and give the organic product. Unsaturated organic groups couple more easily in part because the add readily. The intermediates are also less prone to beta-hydride elimination
.
In one computational study, unsaturated organic groups were shown to undergo much easier coupling reaction on the metal center. The rates for reductive elimination followed the following order:
vinyl-vinyl > phenyl-phenyl > alkynyl-alkynyl > alkyl-alkyl.
The activation barriers and the reaction energies for unsymmetrical R-R′ couplings were found to be close to the averages of the corresponding values of the symmetrical R-R and R′-R′ coupling reactions; for example: vinyl-vinyl > vinyl-alkyl > alkyl-alkyl.
Another mechanistic approach proposes that specifically in aqueous solutions, coupling actually occurs via a radical mechanism rather than a metal-assisted one.
, but some processes often use nickel and copper. A common catalyst is tetrakis(triphenylphosphine)palladium(0)
. Palladium catalysed reactions have several advantages including functional group
tolerance, low sensitivity of organopalladium
compounds towards water and air.
Reviews have been written for example on cobalt, palladium and nickel mediated reactions and on applications
X in the organic partner is usually bromide
, iodide
or triflate
. Ideal leaving groups are chloride, since organic chlorides are cheaper than related compounds. The main group metal in the organometallic partner usually is tin
, zinc
, or boron
.
. Another example of coupling in aqueous media, with the main reacting agent being trimolybdenum-alkylidyne clusters, is that of Bogoslavsky et al.. In general, the oxygen
in the air is more able to disrupt coupling reactions, because many of these reactions occur via unsaturated metal complexes that do not have 18 valence electrons. For example, in nickel
and palladium
cross couplings, a zerovalent complex with two vacant sites (or labile ligands) reacts with the carbon halogen bond to form a metal halogen and a metal carbon bond. Such a zerovalent complex with labile ligands or empty coordination sites is normally very reactive toward oxygen.
not only sat on a shelf for 30 years without any sign of degradation but also decomposed
in water to generate 2-butyne, which is the coupling adduct of its two ethylidyne ligand
s. This, according to the researchers, opens another way for aqueous organometallic chemistry.
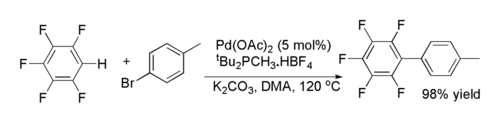
One method for palladium-catalyzed cross-coupling reactions of aryl halides
with fluorinated arenes was reported by Keith Fagnou
and co-workers. It is unusual in that it involves C-H functionalisation at an electron deficient
arene.
Organic chemistry
Organic chemistry is a subdiscipline within chemistry involving the scientific study of the structure, properties, composition, reactions, and preparation of carbon-based compounds, hydrocarbons, and their derivatives...
is a catch-all term for a variety of reactions where two hydrocarbon
Hydrocarbon
In organic chemistry, a hydrocarbon is an organic compound consisting entirely of hydrogen and carbon. Hydrocarbons from which one hydrogen atom has been removed are functional groups, called hydrocarbyls....
fragments are coupled with the aid of a metal catalyst. In one important reaction type a main group organometallic compound of the type RM (R = organic fragment, M = main group centre) reacts with an organic halide of the type R'X with formation of a new carbon-carbon bond
Carbon-carbon bond
A carbon–carbon bond is a covalent bond between two carbon atoms. The most common form is the single bond: a bond composed of two electrons, one from each of the two atoms. The carbon–carbon single bond is a sigma bond and is said to be formed between one hybridized orbital from each...
in the product R-R' Contributions to coupling reactions by Ei-ichi Negishi
Ei-ichi Negishi
is a Japanese chemist who has spent most of his career at Purdue University, United States. He is best known for his discovery of the Negishi coupling. He was awarded the 2010 Nobel Prize in Chemistry "for palladium catalyzed cross couplings in organic synthesis" jointly with Richard F. Heck and...
and Akira Suzuki
Akira Suzuki (chemist)
is a Japanese chemist and Nobel Prize Laureate , who first published the Suzuki reaction, the organic reaction of an aryl- or vinyl-boronic acid with an aryl- or vinyl-halide catalyzed by a palladium complex, in 1979.-Life:...
were recognized with the 2010 Nobel Prize in Chemistry, which was shared with Richard F. Heck
Richard F. Heck
Richard Fred Heck is an American chemist noted for the discovery and development of the Heck reaction, which uses palladium to catalyze organic chemical reactions that couple aryl halides with alkenes....
.
Broadly speaking, two types of coupling reactions are recognized:
- cross couplings involve reactions between two different partners, for example bromobenzene (PhBr) and vinyl chlorideVinyl chlorideVinyl chloride is the organochloride with the formula H2C:CHCl. It is also called vinyl chloride monomer, VCM or chloroethene. This colorless compound is an important industrial chemical chiefly used to produce the polymer polyvinyl chloride . At ambient pressure and temperature, vinyl chloride...
to give styrene (PhCH=CH2). - homocouplings couple two identical partners, for example, the conversion of iodobenzeneIodobenzeneIodobenzene is an organic compound consisting of a benzene ring substitituted with one iodine atom. It is useful as a synthetic intermediate in organic chemistry.-Preparation:...
(PhI) to biphenylBiphenylBiphenyl is an organic compound that forms colorless crystals. It has a distinctively pleasant smell. Biphenyl is an aromatic hydrocarbon with a molecular formula 2...
(Ph-Ph).
Mechanism
The reaction mechanismReaction mechanism
In chemistry, a reaction mechanism is the step by step sequence of elementary reactions by which overall chemical change occurs.Although only the net chemical change is directly observable for most chemical reactions, experiments can often be designed that suggest the possible sequence of steps in...
usually begins with oxidative addition
Oxidative addition
Oxidative addition and reductive elimination are two important and related classes of reactions in organometallic chemistry. Oxidative addition is a process that increases both the oxidation state and coordination number of a metal centre...
of one organic halide to the catalyst. Subsequently, the second partner undergoes transmetallation, which places both coupling partners on the same metal centre. The final step is reductive elimination of the two coupling fragments to regenerate the catalyst and give the organic product. Unsaturated organic groups couple more easily in part because the add readily. The intermediates are also less prone to beta-hydride elimination
Beta-hydride elimination
Beta-hydride elimination is a reaction in which an alkyl group bonded to a metal centre is converted into the corresponding metal-bonded hydride and an alkene. The alkyl must have hydrogens on the beta carbon. For instance butyl groups can undergo this reaction but methyl groups cannot...
.
In one computational study, unsaturated organic groups were shown to undergo much easier coupling reaction on the metal center. The rates for reductive elimination followed the following order:
vinyl-vinyl > phenyl-phenyl > alkynyl-alkynyl > alkyl-alkyl.
The activation barriers and the reaction energies for unsymmetrical R-R′ couplings were found to be close to the averages of the corresponding values of the symmetrical R-R and R′-R′ coupling reactions; for example: vinyl-vinyl > vinyl-alkyl > alkyl-alkyl.
Another mechanistic approach proposes that specifically in aqueous solutions, coupling actually occurs via a radical mechanism rather than a metal-assisted one.
Catalysts
The most popular metal catalyst is palladiumPalladium
Palladium is a chemical element with the chemical symbol Pd and an atomic number of 46. It is a rare and lustrous silvery-white metal discovered in 1803 by William Hyde Wollaston. He named it after the asteroid Pallas, which was itself named after the epithet of the Greek goddess Athena, acquired...
, but some processes often use nickel and copper. A common catalyst is tetrakis(triphenylphosphine)palladium(0)
Tetrakis(triphenylphosphine)palladium(0)
Tetrakispalladium is the chemical compound Pd[P3]4, often abbreviated Pd4, or even PdP4. It is a bright yellow crystalline solid that becomes brown upon decomposition in air.-Structure and properties:...
. Palladium catalysed reactions have several advantages including functional group
Functional group
In organic chemistry, functional groups are specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules. The same functional group will undergo the same or similar chemical reaction regardless of the size of the molecule it is a part of...
tolerance, low sensitivity of organopalladium
Organopalladium
Organopalladium chemistry is a branch of organometallic chemistry that deals with organic palladium compounds and their reactions. Palladium is often used as a catalyst in the reduction of alkenes and alkynes with hydrogen. This process involves the formation of a palladium-carbon covalent bond...
compounds towards water and air.
Reviews have been written for example on cobalt, palladium and nickel mediated reactions and on applications
Leaving groups
The leaving groupLeaving group
In chemistry, a leaving group is a molecular fragment that departs with a pair of electrons in heterolytic bond cleavage. Leaving groups can be anions or neutral molecules. Common anionic leaving groups are halides such as Cl−, Br−, and I−, and sulfonate esters, such as para-toluenesulfonate...
X in the organic partner is usually bromide
Bromide
A bromide is a chemical compound containing bromide ion, that is bromine atom with effective charge of −1. The class name can include ionic compounds such as caesium bromide or covalent compounds such as sulfur dibromide.-Natural occurrence:...
, iodide
Iodide
An iodide ion is the ion I−. Compounds with iodine in formal oxidation state −1 are called iodides. This page is for the iodide ion and its salts. For information on organoiodides, see organohalides. In everyday life, iodide is most commonly encountered as a component of iodized salt,...
or triflate
Triflate
Trifluoromethanesulfonate, also known by the trivial name triflate, is a functional group with the formula CF3SO3-. The triflate group is often represented by -OTf, as opposed to -Tf...
. Ideal leaving groups are chloride, since organic chlorides are cheaper than related compounds. The main group metal in the organometallic partner usually is tin
Tin
Tin is a chemical element with the symbol Sn and atomic number 50. It is a main group metal in group 14 of the periodic table. Tin shows chemical similarity to both neighboring group 14 elements, germanium and lead and has two possible oxidation states, +2 and the slightly more stable +4...
, zinc
Zinc
Zinc , or spelter , is a metallic chemical element; it has the symbol Zn and atomic number 30. It is the first element in group 12 of the periodic table. Zinc is, in some respects, chemically similar to magnesium, because its ion is of similar size and its only common oxidation state is +2...
, or boron
Boron
Boron is the chemical element with atomic number 5 and the chemical symbol B. Boron is a metalloid. Because boron is not produced by stellar nucleosynthesis, it is a low-abundance element in both the solar system and the Earth's crust. However, boron is concentrated on Earth by the...
.
Operating conditions
While many coupling reactions involve reagents that are extremely susceptible to presence of water or oxygen, it is unreasonable to assume that all coupling reactions need to be performed with strict exclusion of water. It is possible to perform palladium-based coupling reactions in aqueous solutions using the water-soluble sulfonated phosphines made by the reaction of triphenyl phosphine with sulfuric acidSulfuric acid
Sulfuric acid is a strong mineral acid with the molecular formula . Its historical name is oil of vitriol. Pure sulfuric acid is a highly corrosive, colorless, viscous liquid. The salts of sulfuric acid are called sulfates...
. Another example of coupling in aqueous media, with the main reacting agent being trimolybdenum-alkylidyne clusters, is that of Bogoslavsky et al.. In general, the oxygen
Oxygen
Oxygen is the element with atomic number 8 and represented by the symbol O. Its name derives from the Greek roots ὀξύς and -γενής , because at the time of naming, it was mistakenly thought that all acids required oxygen in their composition...
in the air is more able to disrupt coupling reactions, because many of these reactions occur via unsaturated metal complexes that do not have 18 valence electrons. For example, in nickel
Nickel
Nickel is a chemical element with the chemical symbol Ni and atomic number 28. It is a silvery-white lustrous metal with a slight golden tinge. Nickel belongs to the transition metals and is hard and ductile...
and palladium
Palladium
Palladium is a chemical element with the chemical symbol Pd and an atomic number of 46. It is a rare and lustrous silvery-white metal discovered in 1803 by William Hyde Wollaston. He named it after the asteroid Pallas, which was itself named after the epithet of the Greek goddess Athena, acquired...
cross couplings, a zerovalent complex with two vacant sites (or labile ligands) reacts with the carbon halogen bond to form a metal halogen and a metal carbon bond. Such a zerovalent complex with labile ligands or empty coordination sites is normally very reactive toward oxygen.
Coupling types
Coupling reactions include (not exhaustive):| Reaction | year | Reactant A | Reactant B | homo/cross | catalyst | remark | |||
| Wurtz reaction Wurtz reaction The Wurtz reaction, named after Charles-Adolphe Wurtz, is a coupling reaction in organic chemistry, organometallic chemistry and recently inorganic main group polymers, whereby two alkyl halides are reacted with sodium to form a new carbon-carbon bond:... |
1855 | R-X | sp³ | R-X | sp³ | homo | Na | ||
| Glaser coupling | 1869 | RC≡CH | sp | RC≡CH | sp | homo | Cu | O2 as H-acceptor | |
| Ullmann reaction Ullmann reaction The Ullmann reaction or Ullmann coupling is a coupling reaction between aryl halides with copper. The reaction is named after Fritz Ullmann.... |
1901 | Ar-X | sp² | Ar-X | sp² | homo | Cu | high temperatures | |
| Gomberg-Bachmann reaction Gomberg-Bachmann reaction The Gomberg–Bachmann reaction, named for the Ukrainian-American chemist Moses Gomberg and the American chemist Werner Emmanuel Bachmann, is an aryl-aryl coupling reaction via a diazonium salt.... |
1924 | Ar-H | sp² | Ar-N2X | sp² | homo | requires base | ||
| Cadiot-Chodkiewicz coupling Cadiot-Chodkiewicz coupling The Cadiot-Chodkiewicz coupling in organic chemistry is a coupling reaction between a terminal alkyne and a haloalkyne catalyzed by a copper salt such as copper bromide and an amine base. The reaction product is a di-acetylene or di-alkyne.... |
1957 | RC≡CH | sp | RC≡CX | sp | cross | Cu | requires base | |
| Castro-Stephens coupling Castro-Stephens coupling The Castro-Stephens Coupling is a cross coupling reaction between a copper acetylide and an aryl halide forming a disubstituted alkyne and a copper halide.... |
1963 | RC≡CH | sp | Ar-X | sp² | cross | Cu | ||
| Gilman reagent Gilman reagent A Gilman reagent is a lithium and copper reagent compound, R2CuLi, where R is an organic radical. These are useful because they react with organic chlorides, bromides, and iodides to replace the halide group with an R group. This is extremely useful in creating larger molecules from smaller... coupling |
1967 | R2CuLi | R-X | cross | |||||
| Cassar reaction | 1970 | Alkene | sp² | R-X | sp³ | cross | Pd | requires base | |
| Kumada coupling Kumada coupling A Kumada coupling or Kumada-Corriu coupling is a cross coupling reaction in organic chemistry between an alkyl or aryl Grignard reagent and an aryl or vinyl halocarbon catalysed by nickel or palladium. This reaction is relevant to organic synthesis because it gives access to styrene derivatives... |
1972 | Ar-MgBr | sp², sp³ | Ar-X | sp² | cross | Pd or Ni | ||
| Heck reaction Heck reaction The Heck reaction is the chemical reaction of an unsaturated halide with an alkene and a base and palladium catalyst to form a substituted alkene. Together with the other palladium-catalyzed cross-coupling reactions, this reaction is of great importance, as it allows one to do substitution... |
1972 | alkene | sp² | R-X | sp² | cross | Pd | requires base | |
| Sonogashira coupling Sonogashira coupling In organic chemistry, a Sonogashira coupling is a coupling reaction of terminal alkynes with aryl or vinyl halides. This reaction was first reported by Kenkichi Sonogashira and Nobue Hagihara in 1975.-Catalyst:... |
1975 | RC≡CH | sp | R-X | sp³ sp² | cross | Pd and Cu | requires base | |
| Negishi coupling Negishi coupling The Negishi coupling is a cross coupling reaction in organic chemistry involving an organozinc compound, an organic halide and a nickel or palladium catalyst creating a new carbon-carbon covalent bond:* The halide X can be chloride, bromine or iodine but also a triflate or acetyloxy group with as... |
1977 | R-Zn-X | sp³, sp², sp | R-X | sp³ sp² | cross | Pd or Ni | ||
| Stille cross coupling | 1978 | R-SnR3 | sp³, sp², sp | R-X | sp³ sp² | cross | Pd | ||
| Suzuki reaction Suzuki reaction The Suzuki reaction is the organic reaction of an aryl- or vinyl-boronic acid with an aryl- or vinyl-halide catalyzed by a palladium complex. It is widely used to synthesize poly-olefins, styrenes, and substituted biphenyls, and has been extended to incorporate alkyl bromides... |
1979 | R-B(OR)2 | sp² | R-X | sp³ sp² | cross | Pd | requires base | |
| Hiyama coupling Hiyama coupling In organic chemistry, a Hiyama coupling is a palladium or nickel-catalyzed cross coupling reaction of organosilanes with organic halides or triflates. Hiyama couplings were first reported by Yasuo Hatanaka and Tamejiro Hiyama in 1988.... |
1988 | R-SiR3 | sp² | R-X | sp³ sp² | cross | Pd | requires base | |
| Buchwald-Hartwig reaction Buchwald-Hartwig reaction The Buchwald–Hartwig reaction is a chemical reaction used in organic chemistry for the synthesis of carbon–nitrogen bonds via the palladium-catalyzed cross-coupling of amines with aryl halides. Though publications with similar focus were published as early as 1983, credit for its development is... |
1994 | R2N-R SnR3 | sp | R-X | sp² | cross | Pd | N-C coupling, second generation free amine | |
| Fukuyama coupling Fukuyama coupling The Fukuyama coupling is a coupling reaction taking place between a thioester and an organozinc halide in the presence of a palladium catalyst. The reaction product is a ketone. This reaction was discovered by Tohru Fukuyama et al. in 1998... |
1998 | RCO(SEt) | sp2 | R-Zn-I | sp3 | cross | Pd | ||
| Coupling reaction overview. For references consult satellite pages | |||||||||
Miscellaneous reactions
In one study, an unusual coupling reaction was described in which an organomolybdenum compound, [Mo3(CCH3)2(OAc)6(H2O)3](CF3SO3)2not only sat on a shelf for 30 years without any sign of degradation but also decomposed
Chemical decomposition
Chemical decomposition, analysis or breakdown is the separation of a chemical compound into elements or simpler compounds. It is sometimes defined as the exact opposite of a chemical synthesis. Chemical decomposition is often an undesired chemical reaction...
in water to generate 2-butyne, which is the coupling adduct of its two ethylidyne ligand
Ligand
In coordination chemistry, a ligand is an ion or molecule that binds to a central metal atom to form a coordination complex. The bonding between metal and ligand generally involves formal donation of one or more of the ligand's electron pairs. The nature of metal-ligand bonding can range from...
s. This, according to the researchers, opens another way for aqueous organometallic chemistry.
One method for palladium-catalyzed cross-coupling reactions of aryl halides
Halogenoarene
In organic chemistry, a halogenoarene, haloarene, or aryl halide, is an organic compound in which a halogen atom is bonded to a carbon atom which is part of an aromatic ring. The haloarene are studied separately from haloalkanes because they exhibit many differences in methods of preparation and...
with fluorinated arenes was reported by Keith Fagnou
Keith Fagnou
Keith Fagnou was a Canadian organic chemist and associate professor of chemistry at the University of Ottawa and a rising star in the field of organic chemistry...
and co-workers. It is unusual in that it involves C-H functionalisation at an electron deficient
Electron deficiency
Electron deficiency occurs when a compound has too few valence electrons for the connections between atoms to be described as covalent bonds. Electron deficient bonds are often better described as 3-center-2-electron bonds...
arene.


