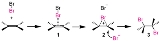
Electrophile
Encyclopedia
In general electrophiles are positively charged species that are attracted to an electron rich centre. In chemistry
, an electrophile (literally electron-lover) is a reagent
attracted to electron
s that participates in a chemical reaction by accepting an electron pair
in order to bond
to a nucleophile
. Because electrophiles accept electrons, they are Lewis acid
s (see acid-base reaction theories
). Most electrophiles are positively charge
d, have an atom that carries a partial positive charge, or have an atom that does not have an octet of electrons.
The electrophiles attack the most electron-populated part of a nucleophile
. The electrophiles frequently seen in the organic syntheses
are cations such as H+
and NO+
, polarized neutral molecules such as HCl
, alkyl halides, acyl halide
s, and carbonyl compounds, polarizable neutral molecules such as Cl2
and Br2
, oxidizing agent
s such as organic peracids, chemical species that do not satisfy the octet rule
such as carbene
s and radicals
, and some lewis acids such as BH3
and DIBAL
.
is one of the three main forms of reaction concerning alkenes.
s. Common reactions include use of bromine water to titrate
against a sample to deduce the number of double bonds present. For example, ethene + bromine
→ 1,2-dibromoethane
:
This takes the form of 3 main steps shown below;
This process is called AdE2 mechanism. Iodine
(I2), chlorine
(Cl2), sulfenyl ion (RS+), mercury
cation (Hg2+), and dichlorocarbene
(:CCl2) also react through similar pathways. The direct conversion of 1 to 3 will appear when the Br− is large excess in the reaction medium. A β-bromo carbenium ion
intermediate may be predominant instead of 3 if the alkene has a cation-stabilizing substituent like phenyl group. There is an example of the isolation of the bromonium ion 2.
. For example, the reaction of HCl with ethylene furnishes chloroethane. The reaction proceeds with a cation intermediate, being different from the above halogen addition. An example is shown below:
In this manner, the stereoselectivity
of the product, that is, from which side Cl− will attack relies on the types of alkenes applied and conditions of the reaction. At least, which of the two carbon atoms will be attacked by H+ is usually decided by Markovnikov's rule
. Thus, H+ attacks the carbon atom that carries fewer substituents so as the more stabilized carbocation (with the more stabilizing substituents) will form.
This process is called A-SE2 mechanism. Hydrogen fluoride
(HF) and hydrogen iodide (HI) react with alkenes in a similar manner, and Markovnikov-type products will be given. Hydrogen bromide (HBr) also takes this pathway, but sometimes a radical process competes and a mixture of isomers may form.
s utilises sulfuric acid
as a catalyst. This reaction occurs in a similar way to the addition reaction but has an extra step in which the OSO3H group is replaced by an OH group, forming an alcohol:
As can be seen, the H2SO4 does take part in the overall reaction, however it remains unchanged so is classified as a catalyst.
This is the reaction in more detail:
Overall, this process adds a molecule of water to a molecule of ethene.
This is an important reaction in industry, as it produces ethanol
, whose purposes include fuels and starting material for other chemicals.
Several methods exist to rank electrophiles in order of reactivity and one of them is devised by Robert Parr
with the electrophilicity index ω
given as:

with the electronegativity
the electronegativity
and chemical hardness. This equation is related to classical equation for electrical power
chemical hardness. This equation is related to classical equation for electrical power
:

where is the resistance
is the resistance
(Ohm
or Ω) and is voltage
is voltage
. In this sense the electrophilicity index is a kind of electrophilic power. Correlations have been found between electrophilicity of various chemical compounds and reaction rates in biochemical systems and such phenomena as allergic contact dermititis.
An electrophilicity index also exists for free radicals. Strongly electrophilic radicals such as the halogens react with electron-rich reaction sites, and strongly nucleophilic radicals such as the 2-hydroxypropyl-2-yl and tert-butyl radical react with a preference for electron-poor reaction sites.
s. These compounds were first described by George A. Olah. Superelectrophiles form as a doubly electron deficient superelectrophile by protosolvation of a cationic electrophile. As observed by Olah, a mixture of acetic acid
and boron trifluoride
is able to remove a hydride ion from isobutane
when combined with hydrofluoric acid
via the formation of a superacid
from BF3 and HF. The responsible reactive intermediate
is the CH3CO2H3 dication. Likewise, methane
can be nitrated to nitromethane
with nitronium tetrafluoroborate
NOBF only in presence of a strong acid like fluorosulfuric acid
.
In gitionic (gitonic) superelectrophiles, charged centers are separated by no more than one atom, for example, the protonitronium ion O=N+=O+—H (a protonated nitronium ion
). And, in distonic superelectrophiles, they are separated by 2 or more atoms, for example, in the fluorination reagent F-TEDA-BF4
Chemistry
Chemistry is the science of matter, especially its chemical reactions, but also its composition, structure and properties. Chemistry is concerned with atoms and their interactions with other atoms, and particularly with the properties of chemical bonds....
, an electrophile (literally electron-lover) is a reagent
Reagent
A reagent is a "substance or compound that is added to a system in order to bring about a chemical reaction, or added to see if a reaction occurs." Although the terms reactant and reagent are often used interchangeably, a reactant is less specifically a "substance that is consumed in the course of...
attracted to electron
Electron
The electron is a subatomic particle with a negative elementary electric charge. It has no known components or substructure; in other words, it is generally thought to be an elementary particle. An electron has a mass that is approximately 1/1836 that of the proton...
s that participates in a chemical reaction by accepting an electron pair
Electron pair
In chemistry, an electron pair consists of two electrons that occupy the same orbital but have opposite spins.Because electrons are fermions, the Pauli exclusion principle forbids these particles from having exactly the same quantum numbers. Therefore the only way to occupy the same orbital, i.e....
in order to bond
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electromagnetic force attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction...
to a nucleophile
Nucleophile
A nucleophile is a species that donates an electron-pair to an electrophile to form a chemical bond in a reaction. All molecules or ions with a free pair of electrons can act as nucleophiles. Because nucleophiles donate electrons, they are by definition Lewis bases.Nucleophilic describes the...
. Because electrophiles accept electrons, they are Lewis acid
Lewis acid
]The term Lewis acid refers to a definition of acid published by Gilbert N. Lewis in 1923, specifically: An acid substance is one which can employ a lone pair from another molecule in completing the stable group of one of its own atoms. Thus, H+ is a Lewis acid, since it can accept a lone pair,...
s (see acid-base reaction theories
Acid-base reaction theories
An acid–base reaction is a chemical reaction that occurs between an acid and a base. Several concepts that provide alternative definitions for the reaction mechanisms involved and their application in solving related problems exist...
). Most electrophiles are positively charge
Electric charge
Electric charge is a physical property of matter that causes it to experience a force when near other electrically charged matter. Electric charge comes in two types, called positive and negative. Two positively charged substances, or objects, experience a mutual repulsive force, as do two...
d, have an atom that carries a partial positive charge, or have an atom that does not have an octet of electrons.
The electrophiles attack the most electron-populated part of a nucleophile
Nucleophile
A nucleophile is a species that donates an electron-pair to an electrophile to form a chemical bond in a reaction. All molecules or ions with a free pair of electrons can act as nucleophiles. Because nucleophiles donate electrons, they are by definition Lewis bases.Nucleophilic describes the...
. The electrophiles frequently seen in the organic syntheses
Organic synthesis
Organic synthesis is a special branch of chemical synthesis and is concerned with the construction of organic compounds via organic reactions. Organic molecules can often contain a higher level of complexity compared to purely inorganic compounds, so the synthesis of organic compounds has...
are cations such as H+
Hydrogen ion
Hydrogen ion is recommended by IUPAC as a general term for all ions of hydrogen and its isotopes.Depending on the charge of the ion, two different classes can be distinguished: positively charged ions and negatively charged ions....
and NO+
Nitrosonium
The nitrosonium ion is NO+, in which the nitrogen atom is bonded to an oxygen atom with a bond order of 3, and the overall diatomic species bears a positive charge. This ion is usually obtained as the following salts: NOClO4, NOSO4H , and NOBF4. The ClO and BF salts are slightly soluble in CH3CN...
, polarized neutral molecules such as HCl
Hydrogen chloride
The compound hydrogen chloride has the formula HCl. At room temperature, it is a colorless gas, which forms white fumes of hydrochloric acid upon contact with atmospheric humidity. Hydrogen chloride gas and hydrochloric acid are important in technology and industry...
, alkyl halides, acyl halide
Acyl halide
An acyl halide is a chemical compound derived from an oxoacid by replacing a hydroxyl group with a halide group....
s, and carbonyl compounds, polarizable neutral molecules such as Cl2
Chlorine
Chlorine is the chemical element with atomic number 17 and symbol Cl. It is the second lightest halogen, found in the periodic table in group 17. The element forms diatomic molecules under standard conditions, called dichlorine...
and Br2
Bromine
Bromine ") is a chemical element with the symbol Br, an atomic number of 35, and an atomic mass of 79.904. It is in the halogen element group. The element was isolated independently by two chemists, Carl Jacob Löwig and Antoine Jerome Balard, in 1825–1826...
, oxidizing agent
Oxidizing agent
An oxidizing agent can be defined as a substance that removes electrons from another reactant in a redox chemical reaction...
s such as organic peracids, chemical species that do not satisfy the octet rule
Octet rule
The octet rule is a chemical rule of thumb that states that atoms of low The octet rule is a chemical rule of thumb that states that atoms of low The octet rule is a chemical rule of thumb that states that atoms of low (The octet rule is a chemical rule of thumb that states that atoms of low (...
such as carbene
Carbene
In chemistry, a carbene is a molecule containing a neutral carbon atom with a valence of two and two unshared valence electrons. The general formula is RR'C:, but the carbon can instead be double-bonded to one group. The term "carbene" may also merely refer to the compound H2C:, also called...
s and radicals
Radical (chemistry)
Radicals are atoms, molecules, or ions with unpaired electrons on an open shell configuration. Free radicals may have positive, negative, or zero charge...
, and some lewis acids such as BH3
Borane
In chemistry, a borane is a chemical compound of boron and hydrogen. The boranes comprise a large group of compounds with the generic formulae of BxHy. These compounds do not occur in nature. Many of the boranes readily oxidise on contact with air, some violently. The parent member BH3 is called...
and DIBAL
Diisobutylaluminium hydride
Diisobutylaluminium hydride, DIBAL, DIBAL-H or DIBAH, is a reducing agent with the formula 2, where i-Bu represents isobutyl...
.
Alkenes
Electrophilic additionElectrophilic addition
In organic chemistry, an electrophilic addition reaction is an addition reaction where, in a chemical compound, a π bond is broken and two new σ bonds are formed...
is one of the three main forms of reaction concerning alkenes.
- HydrogenationHydrogenationHydrogenation, to treat with hydrogen, also a form of chemical reduction, is a chemical reaction between molecular hydrogen and another compound or element, usually in the presence of a catalyst. The process is commonly employed to reduce or saturate organic compounds. Hydrogenation typically...
by the addition of hydrogen over the double bond - Electrophilic additionElectrophilic additionIn organic chemistry, an electrophilic addition reaction is an addition reaction where, in a chemical compound, a π bond is broken and two new σ bonds are formed...
reactions with halogens and sulfuric acid - HydrationHydration reactionIn organic chemistry, a hydration reaction is a chemical reaction in which a hydroxyl group and a hydrogen cation are added to the two carbon atoms bonded together in the carbon-carbon double bond which makes up an alkene functional group. The reaction usually runs in a strong acidic, aqueous...
to form alcohols.
Addition of halogens
These occur between alkenes and electrophiles, often halogens as in halogen addition reactionHalogen addition reaction
A halogen addition reaction is a simple organic reaction where a halogen molecule is added to the carbon–carbon double bond of an alkene functional group.The general chemical formula of the halogen addition reaction is:...
s. Common reactions include use of bromine water to titrate
Titration
Titration, also known as titrimetry, is a common laboratory method of quantitative chemical analysis that is used to determine the unknown concentration of an identified analyte. Because volume measurements play a key role in titration, it is also known as volumetric analysis. A reagent, called the...
against a sample to deduce the number of double bonds present. For example, ethene + bromine
Bromine
Bromine ") is a chemical element with the symbol Br, an atomic number of 35, and an atomic mass of 79.904. It is in the halogen element group. The element was isolated independently by two chemists, Carl Jacob Löwig and Antoine Jerome Balard, in 1825–1826...
→ 1,2-dibromoethane
1,2-Dibromoethane
1,2-Dibromoethane, also known as ethylene dibromide , is the chemical compound with the formula BrCH2CH2Br. Although trace amounts occur naturally in the ocean, where it is formed probably by algae and kelp, it is mainly a synthetic...
:
- C2H4 + Br2 → BrCH2CH2Br
This takes the form of 3 main steps shown below;
- Forming of a π-complex
- The electrophilic Br-Br molecule interacts with electron-rich alkene molecule to form a π-complex 1.
- Forming of a three-membered bromonium ion
- The alkene is working as an electron donor and bromine as an electrophile. The three-membered bromonium ion 2 consisted with two carbon atoms and a bromine atom forms with a release of Br−.
- Attacking of bromide ion
- Forming of a π-complex
-
- The bromonium ion is opened by the attack of Br− from the back side. This yields the vicinalVicinal (chemistry)In chemistry vicinal stands for any two functional groups bonded to two adjacent carbon atoms. For example the molecule 2,3-dibromobutane carries two vicinal bromine atoms and 1,3-dibromobutane does not....
dibromide with an antiperiplanar configuration. When other nucleophiles such as water or alcohol are existing, these may attack 2 to give an alcohol or an ether.
- The bromonium ion is opened by the attack of Br− from the back side. This yields the vicinal
This process is called AdE2 mechanism. Iodine
Iodine
Iodine is a chemical element with the symbol I and atomic number 53. The name is pronounced , , or . The name is from the , meaning violet or purple, due to the color of elemental iodine vapor....
(I2), chlorine
Chlorine
Chlorine is the chemical element with atomic number 17 and symbol Cl. It is the second lightest halogen, found in the periodic table in group 17. The element forms diatomic molecules under standard conditions, called dichlorine...
(Cl2), sulfenyl ion (RS+), mercury
Mercury (element)
Mercury is a chemical element with the symbol Hg and atomic number 80. It is also known as quicksilver or hydrargyrum...
cation (Hg2+), and dichlorocarbene
Dichlorocarbene
Dichlorocarbene is a carbene commonly encountered in organic chemistry. This reactive intermediate with chemical formula CCl2 is easily available by reaction of chloroform and a base such as potassium t-butoxide or sodium hydroxide dissolved in water...
(:CCl2) also react through similar pathways. The direct conversion of 1 to 3 will appear when the Br− is large excess in the reaction medium. A β-bromo carbenium ion
Carbenium ion
A carbenium ion is a carbocation of the trivalent and classical type R3C+. It is one of two types of carbocation, the other being a carbonium ion. In older literature a carbocation of the type R3C+ may still be referred to as a carbonium ion, a term that is used now for five-coordinate carbon...
intermediate may be predominant instead of 3 if the alkene has a cation-stabilizing substituent like phenyl group. There is an example of the isolation of the bromonium ion 2.
Addition of hydrogen halides
Hydrogen halides such as hydrogen chloride (HCl) adds to alkenes to give alkyl halide in hydrohalogenationHydrohalogenation
A hydrohalogenation reaction is the electrophilic addition of hydrohalic acids like hydrogen chloride or hydrogen bromide to alkenes to yield the corresponding haloalkanes....
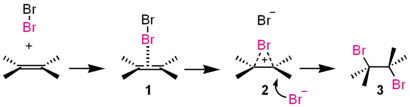
. For example, the reaction of HCl with ethylene furnishes chloroethane. The reaction proceeds with a cation intermediate, being different from the above halogen addition. An example is shown below:
- Proton (H+) adds (by working as an electrophile) to one of the carbon atoms on the alkene to form cation 1.
- Chloride ion (Cl−) combines with the cation 1 to form the adducts 2 and 3.
In this manner, the stereoselectivity
Stereoselectivity
In chemistry, stereoselectivity is the property of a chemical reaction in which a single reactant forms an unequal mixture of stereoisomers during the non-stereospecific creation of a new stereocenter or during the non-stereospecific transformation of a pre-existing one...
of the product, that is, from which side Cl− will attack relies on the types of alkenes applied and conditions of the reaction. At least, which of the two carbon atoms will be attacked by H+ is usually decided by Markovnikov's rule
Markovnikov's rule
In organic chemistry, Markovnikov's rule or Markownikoff's rule is an observation based on Zaitsev's rule. It was formulated by the Russian chemist Vladimir Vasilevich Markovnikov in 1870....
. Thus, H+ attacks the carbon atom that carries fewer substituents so as the more stabilized carbocation (with the more stabilizing substituents) will form.
This process is called A-SE2 mechanism. Hydrogen fluoride
Hydrogen fluoride
Hydrogen fluoride is a chemical compound with the formula HF. This colorless gas is the principal industrial source of fluorine, often in the aqueous form as hydrofluoric acid, and thus is the precursor to many important compounds including pharmaceuticals and polymers . HF is widely used in the...
(HF) and hydrogen iodide (HI) react with alkenes in a similar manner, and Markovnikov-type products will be given. Hydrogen bromide (HBr) also takes this pathway, but sometimes a radical process competes and a mixture of isomers may form.
Hydration
One of the more complex hydration reactionHydration reaction
In organic chemistry, a hydration reaction is a chemical reaction in which a hydroxyl group and a hydrogen cation are added to the two carbon atoms bonded together in the carbon-carbon double bond which makes up an alkene functional group. The reaction usually runs in a strong acidic, aqueous...
s utilises sulfuric acid
Sulfuric acid
Sulfuric acid is a strong mineral acid with the molecular formula . Its historical name is oil of vitriol. Pure sulfuric acid is a highly corrosive, colorless, viscous liquid. The salts of sulfuric acid are called sulfates...
as a catalyst. This reaction occurs in a similar way to the addition reaction but has an extra step in which the OSO3H group is replaced by an OH group, forming an alcohol:
- C2H4 + H2O → C2H5OH
As can be seen, the H2SO4 does take part in the overall reaction, however it remains unchanged so is classified as a catalyst.
This is the reaction in more detail:
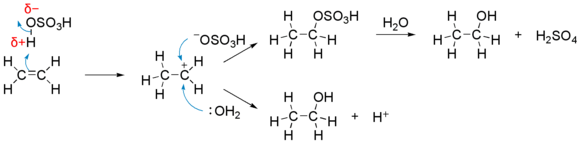
- The H-OSO3H molecule has a δ+ charge on the initial H atom. This is attracted to and reacts with the double bond in the same way as before.
- The remaining (negatively charged) −OSO3H ion then attaches to the carbocationCarbocationA carbocation is an ion with a positively-charged carbon atom. The charged carbon atom in a carbocation is a "sextet", i.e. it has only six electrons in its outer valence shell instead of the eight valence electrons that ensures maximum stability . Therefore carbocations are often reactive,...
, forming ethyl hydrogensulphate (upper way on the above scheme). - When water (H2O) is added and the mixture heated, ethanol (C2H5OH) is produced. The "spare" hydrogen atom from the water goes into "replacing" the "lost" hydrogen and, thus, reproduces sulfuric acid. Another pathway in which water molecule combines directly to the intermediate carbocation (lower way) is also possible. This pathway become predominant when aqueous sulfuric acid is used.
Overall, this process adds a molecule of water to a molecule of ethene.
This is an important reaction in industry, as it produces ethanol
Ethanol
Ethanol, also called ethyl alcohol, pure alcohol, grain alcohol, or drinking alcohol, is a volatile, flammable, colorless liquid. It is a psychoactive drug and one of the oldest recreational drugs. Best known as the type of alcohol found in alcoholic beverages, it is also used in thermometers, as a...
, whose purposes include fuels and starting material for other chemicals.
Electrophilicity scale
| Electrophilicity index | |
| Fluorine Fluorine Fluorine is the chemical element with atomic number 9, represented by the symbol F. It is the lightest element of the halogen column of the periodic table and has a single stable isotope, fluorine-19. At standard pressure and temperature, fluorine is a pale yellow gas composed of diatomic... |
3.86 |
| Chlorine Chlorine Chlorine is the chemical element with atomic number 17 and symbol Cl. It is the second lightest halogen, found in the periodic table in group 17. The element forms diatomic molecules under standard conditions, called dichlorine... |
3.67 |
| Bromine Bromine Bromine ") is a chemical element with the symbol Br, an atomic number of 35, and an atomic mass of 79.904. It is in the halogen element group. The element was isolated independently by two chemists, Carl Jacob Löwig and Antoine Jerome Balard, in 1825–1826... |
3.40 |
| Iodine Iodine Iodine is a chemical element with the symbol I and atomic number 53. The name is pronounced , , or . The name is from the , meaning violet or purple, due to the color of elemental iodine vapor.... |
3.09 |
| Hypochlorite Hypochlorite The hypochlorite ion, also known as chlorate anion is ClO−. A hypochlorite compound is a chemical compound containing this group, with chlorine in oxidation state +1.Hypochlorites are the salts of hypochlorous acid... |
2.52 |
| sulfur dioxide Sulfur dioxide Sulfur dioxide is the chemical compound with the formula . It is released by volcanoes and in various industrial processes. Since coal and petroleum often contain sulfur compounds, their combustion generates sulfur dioxide unless the sulfur compounds are removed before burning the fuel... |
2.01 |
| Carbon disulfide Carbon disulfide Carbon disulfide is a colorless volatile liquid with the formula CS2. The compound is used frequently as a building block in organic chemistry as well as an industrial and chemical non-polar solvent... |
1.64 |
| Benzene Benzene Benzene is an organic chemical compound. It is composed of 6 carbon atoms in a ring, with 1 hydrogen atom attached to each carbon atom, with the molecular formula C6H6.... |
1.45 |
| Sodium Sodium Sodium is a chemical element with the symbol Na and atomic number 11. It is a soft, silvery-white, highly reactive metal and is a member of the alkali metals; its only stable isotope is 23Na. It is an abundant element that exists in numerous minerals, most commonly as sodium chloride... |
0.88 |
| Some selected values (no dimensions) | |
Several methods exist to rank electrophiles in order of reactivity and one of them is devised by Robert Parr
Robert Parr
Robert Ghormley Parr is a theoretical chemist. He is a chemistry professor at the University of North Carolina at Chapel Hill.-Career:...
with the electrophilicity index ω
Omega
Omega is the 24th and last letter of the Greek alphabet. In the Greek numeric system, it has a value of 800. The word literally means "great O" , as opposed to omicron, which means "little O"...
given as:

with
 the electronegativity
the electronegativityElectronegativity
Electronegativity, symbol χ , is a chemical property that describes the tendency of an atom or a functional group to attract electrons towards itself. An atom's electronegativity is affected by both its atomic number and the distance that its valence electrons reside from the charged nucleus...
and
 chemical hardness. This equation is related to classical equation for electrical power
chemical hardness. This equation is related to classical equation for electrical powerElectric power
Electric power is the rate at which electric energy is transferred by an electric circuit. The SI unit of power is the watt.-Circuits:Electric power, like mechanical power, is represented by the letter P in electrical equations...
:

where
 is the resistance
is the resistanceElectrical resistance
The electrical resistance of an electrical element is the opposition to the passage of an electric current through that element; the inverse quantity is electrical conductance, the ease at which an electric current passes. Electrical resistance shares some conceptual parallels with the mechanical...
(Ohm
Ohm
The ohm is the SI unit of electrical resistance, named after German physicist Georg Simon Ohm.- Definition :The ohm is defined as a resistance between two points of a conductor when a constant potential difference of 1 volt, applied to these points, produces in the conductor a current of 1 ampere,...
or Ω) and
 is voltage
is voltageVoltage
Voltage, otherwise known as electrical potential difference or electric tension is the difference in electric potential between two points — or the difference in electric potential energy per unit charge between two points...
. In this sense the electrophilicity index is a kind of electrophilic power. Correlations have been found between electrophilicity of various chemical compounds and reaction rates in biochemical systems and such phenomena as allergic contact dermititis.
An electrophilicity index also exists for free radicals. Strongly electrophilic radicals such as the halogens react with electron-rich reaction sites, and strongly nucleophilic radicals such as the 2-hydroxypropyl-2-yl and tert-butyl radical react with a preference for electron-poor reaction sites.
Superelectrophiles
Superelectrophiles are defined as cationic electrophilic reagents with greatly enhanced reactivities in the presence of superacidSuperacid
According to the classical definition superacid is an acid with an acidity greater than that of 100% pure sulfuric acid, which has a Hammett acidity function of −12. According to the modern definition, superacid is a medium, in which the chemical potential of the proton is higher than in pure...
s. These compounds were first described by George A. Olah. Superelectrophiles form as a doubly electron deficient superelectrophile by protosolvation of a cationic electrophile. As observed by Olah, a mixture of acetic acid
Acetic acid
Acetic acid is an organic compound with the chemical formula CH3CO2H . It is a colourless liquid that when undiluted is also called glacial acetic acid. Acetic acid is the main component of vinegar , and has a distinctive sour taste and pungent smell...
and boron trifluoride
Boron trifluoride
Boron trifluoride is the chemical compound with the formula BF3. This pungent colourless toxic gas forms white fumes in moist air. It is a useful Lewis acid and a versatile building block for other boron compounds.-Structure and bonding:...
is able to remove a hydride ion from isobutane
Isobutane
Isobutane, also known as methylpropane, is an isomer of butane. It is the simplest alkane with a tertiary carbon. Concerns with depletion of the ozone layer by freon gases have led to increased use of isobutane as a gas for refrigeration systems, especially in domestic refrigerators and freezers,...
when combined with hydrofluoric acid
Hydrofluoric acid
Hydrofluoric acid is a solution of hydrogen fluoride in water. It is a valued source of fluorine and is the precursor to numerous pharmaceuticals such as fluoxetine and diverse materials such as PTFE ....
via the formation of a superacid
Superacid
According to the classical definition superacid is an acid with an acidity greater than that of 100% pure sulfuric acid, which has a Hammett acidity function of −12. According to the modern definition, superacid is a medium, in which the chemical potential of the proton is higher than in pure...
from BF3 and HF. The responsible reactive intermediate
Reactive intermediate
In chemistry a reactive intermediate is a short-lived, high energy, highly reactive molecule. When generated in a chemical reaction it will quickly convert into a more stable molecule. Only in exceptional cases can these compounds be isolated and stored, e.g. low temperatures, matrix isolation...
is the CH3CO2H3 dication. Likewise, methane
Methane
Methane is a chemical compound with the chemical formula . It is the simplest alkane, the principal component of natural gas, and probably the most abundant organic compound on earth. The relative abundance of methane makes it an attractive fuel...
can be nitrated to nitromethane
Nitromethane
Nitromethane is an organic compound with the chemical formula . It is the simplest organic nitro compound. It is a slightly viscous, highly polar liquid commonly used as a solvent in a variety of industrial applications such as in extractions, as a reaction medium, and as a cleaning solvent...
with nitronium tetrafluoroborate
Nitronium tetrafluoroborate
Nitronium tetrafluoroborate is an inorganic compound with formula NO2BF4. It is a salt of nitronium cation and tetrafluoroborate anion. It is a colorless crystalline solid, which reacts heavily with water to form the corrosive acids HF and HNO3. As such, it must be handled under water-free conditions...
NOBF only in presence of a strong acid like fluorosulfuric acid
Fluorosulfuric acid
Fluorosulfuric acid is the inorganic compound with the formula HSO3F. It is one of the strongest acids commercially available and is a superacid. The formula HFSO3 emphasizes its relationship to sulfuric acid, H2SO4; HSO3F is a tetrahedral molecule.-Chemical properties:Fluorosulfuric acid is a...
.
In gitionic (gitonic) superelectrophiles, charged centers are separated by no more than one atom, for example, the protonitronium ion O=N+=O+—H (a protonated nitronium ion
Nitronium ion
The nitronium ion, or sometimes the nitryl ion , , is a generally reactive cation created by the removal of an electron from the paramagnetic nitrogen dioxide molecule, or the protonation of nitric acid....
). And, in distonic superelectrophiles, they are separated by 2 or more atoms, for example, in the fluorination reagent F-TEDA-BF4
F-TEDA-BF4
1-Chloromethyl-4-fluoro-1,4-diazoniabicyclo[2.2.2]octane bis or Selectfluor® , a trademark of Air Products and Chemicals, is a reagent in chemistry that is used as a fluorine donor. This compound is a derivative of the heterocycle DABCO®...

