
1,2-rearrangement
Encyclopedia
A 1,2-rearrangement or 1,2-migration or 1,2-shift or Whitmore
1,2-shift is an organic reaction
where a substituent
moves from one atom to another atom in a chemical compound
. In a 1,2 shift the movement involves two adjacent atoms but moves over larger distances are possible. In the example below the substituent R moves from carbon atom C2 to C3.
 The rearrangement is intramolecular
The rearrangement is intramolecular
and the starting compound and reaction product are structural isomers. The 1,2-rearrangement belongs to a broad class of chemical reactions called rearrangement reaction
s.
A rearrangement involving a hydrogen
atom is called a 1,2-hydride shift. If the substituent being rearranged is an alkyl group, it is named according to the alkyl group's anion: i.e. 1,2-methanide shift, 1,2-ethanide shift, etc.
such as:
The driving force for the actual migration of a substituent in step two of the rearrangement is the formation of a more stable intermediate. For instance a tertiary carbocation is more stable than a secondary carbocation and therefore the SN1 reaction
of neopentyl bromide with ethanol yields tert-pentyl ethyl ether.
Carbocation rearrangements are more common than the carbanion or radical counterparts. This observation can be explained on the basis of Hückel's rule
. A cyclic carbocationic transition state
is aromatic and stabilized because it holds 2 electrons. In an anionic transition state on the other hand 4 electrons are present thus antiaromatic and destabilized. A radical transition state is neither stabilized or destabilized.
The most important carbocation 1,2-shift is the Wagner–Meerwein rearrangement. A carbanionic 1,2-shift is involved in the benzilic acid rearrangement
.
in 1911 was the conversion of bis(triphenylmethyl)peroxide 1 to the tetraphenylethane 2.
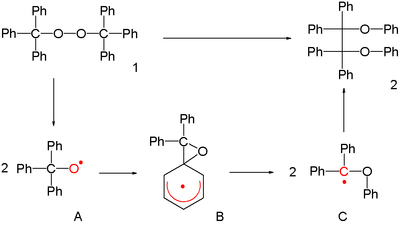 The reaction proceeds through the triphenylmethoxyl radical A, a rearrangement to diphenylphenoxymethyl C and its dimerization. It is unclear to this day whether in this rearrangement the cyclohexadienyl radical intermediate B is a transition state
The reaction proceeds through the triphenylmethoxyl radical A, a rearrangement to diphenylphenoxymethyl C and its dimerization. It is unclear to this day whether in this rearrangement the cyclohexadienyl radical intermediate B is a transition state
or a reactive intermediate
as it (or any other such species) has thus far eluded detection by ESR spectroscopy .
An example of a less common radical 1,2-shift can be found in the gas phase pyrolysis of certain polycyclic aromatic compounds . The energy required in an aryl radical
for the 1,2-shift can be high (up to 60 kcal
/mol
or 250 kJ
/mol) but much less than that required for a proton abstraction to an aryne
(82 kcal/mol or 340 kJ/mol). In alkene
radicals proton abstraction to an alkyne
is preferred.

Frank C. Whitmore
Frank Clifford Whitmore , nicknamed "Rocky", was a prominent chemist who submitted significant evidence for the existence of carbocation mechanisms in organic chemistry.He was born in 1887 in the town of North Attleborough, Massachusetts....
1,2-shift is an organic reaction
Organic reaction
Organic reactions are chemical reactions involving organic compounds. The basic organic chemistry reaction types are addition reactions, elimination reactions, substitution reactions, pericyclic reactions, rearrangement reactions, photochemical reactions and redox reactions. In organic synthesis,...
where a substituent
Substituent
In organic chemistry and biochemistry, a substituent is an atom or group of atoms substituted in place of a hydrogen atom on the parent chain of a hydrocarbon...
moves from one atom to another atom in a chemical compound
Chemical compound
A chemical compound is a pure chemical substance consisting of two or more different chemical elements that can be separated into simpler substances by chemical reactions. Chemical compounds have a unique and defined chemical structure; they consist of a fixed ratio of atoms that are held together...
. In a 1,2 shift the movement involves two adjacent atoms but moves over larger distances are possible. In the example below the substituent R moves from carbon atom C2 to C3.

Intramolecular
Intramolecular in chemistry describes a process or characteristic limited within the structure of a single molecule, a property or phenomenon limited to the extent of a single molecule.- Examples :...
and the starting compound and reaction product are structural isomers. The 1,2-rearrangement belongs to a broad class of chemical reactions called rearrangement reaction
Rearrangement reaction
A rearrangement reaction is a broad class of organic reactions where the carbon skeleton of a molecule is rearranged to give a structural isomer of the original molecule. Often a substituent moves from one atom to another atom in the same molecule...
s.
A rearrangement involving a hydrogen
Hydrogen
Hydrogen is the chemical element with atomic number 1. It is represented by the symbol H. With an average atomic weight of , hydrogen is the lightest and most abundant chemical element, constituting roughly 75% of the Universe's chemical elemental mass. Stars in the main sequence are mainly...
atom is called a 1,2-hydride shift. If the substituent being rearranged is an alkyl group, it is named according to the alkyl group's anion: i.e. 1,2-methanide shift, 1,2-ethanide shift, etc.
Reaction mechanism
A 1,2-rearrangement is often initialised by the formation of a reactive intermediateReactive intermediate
In chemistry a reactive intermediate is a short-lived, high energy, highly reactive molecule. When generated in a chemical reaction it will quickly convert into a more stable molecule. Only in exceptional cases can these compounds be isolated and stored, e.g. low temperatures, matrix isolation...
such as:
- a carbocationCarbocationA carbocation is an ion with a positively-charged carbon atom. The charged carbon atom in a carbocation is a "sextet", i.e. it has only six electrons in its outer valence shell instead of the eight valence electrons that ensures maximum stability . Therefore carbocations are often reactive,...
by heterolysisHeterolysisIn chemistry, heterolysis or heterolytic fission is chemical bond cleavage of a neutral molecule generating a cation and an anion. In this process the two electrons that make up the bond are assigned to the same fragment...
in a nucleophilic rearrangement or anionotropic rearrangement - a carbanionCarbanionA carbanion is an anion in which carbon has an unshared pair of electrons and bears a negative charge usually with three substituents for a total of eight valence electrons. The carbanion exists in a trigonal pyramidal geometry. Formally a carbanion is the conjugate base of a carbon acid.where B...
in a electrophilic rearrangement or cationotropic rearrangement - a free radical by homolysisHomolysisIn general it means breakdown to equal pieces There are separate meanings for the word in chemistry and biology.-Homolysis in chemistry:...
- a nitreneNitreneIn chemistry, a nitrene is the nitrogen analogue of a carbene. The nitrogen atom has only 6 valence electrons and is therefore considered an electrophile...
.
The driving force for the actual migration of a substituent in step two of the rearrangement is the formation of a more stable intermediate. For instance a tertiary carbocation is more stable than a secondary carbocation and therefore the SN1 reaction
SN1 reaction
The SN1 reaction is a substitution reaction in organic chemistry. "SN" stands for nucleophilic substitution and the "1" represents the fact that the rate-determining step is unimolecular...
of neopentyl bromide with ethanol yields tert-pentyl ethyl ether.
Carbocation rearrangements are more common than the carbanion or radical counterparts. This observation can be explained on the basis of Hückel's rule
Hückel's rule
In organic chemistry, Hückel's rule estimates whether a planar ring molecule will have aromatic properties. The quantum mechanical basis for its formulation was first worked out by physical chemist Erich Hückel in 1931...
. A cyclic carbocationic transition state
Transition state
The transition state of a chemical reaction is a particular configuration along the reaction coordinate. It is defined as the state corresponding to the highest energy along this reaction coordinate. At this point, assuming a perfectly irreversible reaction, colliding reactant molecules will always...
is aromatic and stabilized because it holds 2 electrons. In an anionic transition state on the other hand 4 electrons are present thus antiaromatic and destabilized. A radical transition state is neither stabilized or destabilized.
The most important carbocation 1,2-shift is the Wagner–Meerwein rearrangement. A carbanionic 1,2-shift is involved in the benzilic acid rearrangement
Benzilic acid rearrangement
The benzilic acid rearrangement is the rearrangement reaction of benzil with potassium hydroxide to benzilic acid. First performed by Justus Liebig in 1838 this reaction type is displayed by 1,2-diketones in general...
.
Radical 1,2-rearrangements
The first radical 1,2-rearrangement reported by Heinrich Otto WielandHeinrich Otto Wieland
Heinrich Otto Wieland was a German chemist. He won the 1927 Nobel Prize in Chemistry for his research into the bile acids. In 1901 Wieland received his doctorate at the University of Munich while studying under Johannes Thiele...
in 1911 was the conversion of bis(triphenylmethyl)peroxide 1 to the tetraphenylethane 2.

Transition state
The transition state of a chemical reaction is a particular configuration along the reaction coordinate. It is defined as the state corresponding to the highest energy along this reaction coordinate. At this point, assuming a perfectly irreversible reaction, colliding reactant molecules will always...
or a reactive intermediate
Reactive intermediate
In chemistry a reactive intermediate is a short-lived, high energy, highly reactive molecule. When generated in a chemical reaction it will quickly convert into a more stable molecule. Only in exceptional cases can these compounds be isolated and stored, e.g. low temperatures, matrix isolation...
as it (or any other such species) has thus far eluded detection by ESR spectroscopy .
An example of a less common radical 1,2-shift can be found in the gas phase pyrolysis of certain polycyclic aromatic compounds . The energy required in an aryl radical
Aryl radical
An Aryl radical in organic chemistry is an reactive intermediate and an arene compound incorporating one free radical carbon atom as part of the ring structure. As such it is the radical counterpart of the Arenium ion. The parent compound is the phenyl radical C6H5....
for the 1,2-shift can be high (up to 60 kcal
Calorie
The calorie is a pre-SI metric unit of energy. It was first defined by Nicolas Clément in 1824 as a unit of heat, entering French and English dictionaries between 1841 and 1867. In most fields its use is archaic, having been replaced by the SI unit of energy, the joule...
/mol
Mole (unit)
The mole is a unit of measurement used in chemistry to express amounts of a chemical substance, defined as an amount of a substance that contains as many elementary entities as there are atoms in 12 grams of pure carbon-12 , the isotope of carbon with atomic weight 12. This corresponds to a value...
or 250 kJ
Joule
The joule ; symbol J) is a derived unit of energy or work in the International System of Units. It is equal to the energy expended in applying a force of one newton through a distance of one metre , or in passing an electric current of one ampere through a resistance of one ohm for one second...
/mol) but much less than that required for a proton abstraction to an aryne
Aryne
In chemistry, an aryne is an uncharged reactive intermediate derived from an aromatic system by removal of two ortho substituents, leaving two orbitals with two electrons distributed between them....
(82 kcal/mol or 340 kJ/mol). In alkene
Alkene
In organic chemistry, an alkene, olefin, or olefine is an unsaturated chemical compound containing at least one carbon-to-carbon double bond...
radicals proton abstraction to an alkyne
Alkyne
Alkynes are hydrocarbons that have a triple bond between two carbon atoms, with the formula CnH2n-2. Alkynes are traditionally known as acetylenes, although the name acetylene also refers specifically to C2H2, known formally as ethyne using IUPAC nomenclature...
is preferred.

1,2 rearrangements
The following mechanisms involve a 1,2-rearrangement:- 1,2-Wittig rearrangement1,2-Wittig rearrangementA 1,2-Wittig rearrangement is a categorization of chemical reactions in organic chemistry, and consists of a 1,2-rearrangement of an ether with an alkyllithium compound. The reaction is named for Nobel Prize winning chemist Georg Wittig....
- Alpha-ketol rearrangementAlpha-Ketol RearrangementThe α-ketol rearrangement is the acid-, base-, or heat-induced 1,2-migration of an alkyl or aryl group in an α-hydroxy ketone or aldehyde to give an isomeric product.-Introduction:...
- Beckmann rearrangementBeckmann rearrangementThe Beckmann rearrangement, named after the German chemist Ernst Otto Beckmann , is an acid-catalyzed rearrangement of an oxime to an amide...
- Benzilic acid rearrangementBenzilic acid rearrangementThe benzilic acid rearrangement is the rearrangement reaction of benzil with potassium hydroxide to benzilic acid. First performed by Justus Liebig in 1838 this reaction type is displayed by 1,2-diketones in general...
- Brook rearrangementBrook rearrangementThe Brook rearrangement in organic chemistry is a rearrangement reaction in which a organosilyl group switches position with a hydroxyl proton over a carbon to oxygen covalent bond under the influence of a base . It is named for the Canadian chemist Adrian Gibbs Brook...
- Criegee rearrangementCriegee rearrangementThe Criegee rearrangement is a rearrangement reaction named after Rudolf Criegee. In this organic reaction a tertiary alcohol is cleaved in an organic oxidation by a peroxyacid to a ketone...
- Curtius rearrangementCurtius rearrangementThe Curtius rearrangement , as first defined by Theodor Curtius, is a chemical reaction that involves the rearrangement of an acyl azide to an isocyanate. Several reviews have been published....
- Dowd–Beckwith ring expansion reaction
- Favorskii rearrangementFavorskii rearrangementThe Favorskii rearrangement , named for the Russian chemist Alexei Yevgrafovich Favorskii, is most principally a rearrangement of cyclopropanones and α-halo ketones which leads to carboxylic acid derivatives. In the case of cyclic α-halo ketones, the Favorski rearrangement constitutes a ring...
- Friedel–Crafts reaction
- Fritsch–Buttenberg–Wiechell rearrangement
- Halogen dance rearrangementHalogen dance rearrangementThe halogen dance rearrangement is an organic reaction in which a halogen substituent moves to a new position on an aromatic ring system. The reaction belongs to a class of organic reactions called 1,2-rearrangements. The original halogen dance is the base-catalysed rearrangement of...
- Hofmann rearrangementHofmann rearrangementThe Hofmann rearrangement is the organic reaction of a primary amide to a primary amine with one fewer carbon atom.The reaction is named after its discoverer: August Wilhelm von Hofmann...
- Lossen rearrangementLossen rearrangementThe Lossen rearrangement is the conversion of a hydroxamic acid to an isocyanate via the formation of an O-acyl, sulfonyl, or phosphoryl intermediate hydroxamic acid O-derivative and then conversion to its conjugate base. Here, 4-Toluenesulfonyl chloride is used to form a sulfonyl O-derivative...
- Pinacol rearrangementPinacol rearrangementThe pinacol rearrangement or pinacol-pinacolone rearrangement is a method for converting a 1,2-diol to a carbonyl compound in organic chemistry. This 1,2-rearrangement takes place under acidic conditions...
- Seyferth–Gilbert homologation
- SN1 reactionSN1 reactionThe SN1 reaction is a substitution reaction in organic chemistry. "SN" stands for nucleophilic substitution and the "1" represents the fact that the rate-determining step is unimolecular...
(generally) - Stevens rearrangementStevens rearrangementThe Stevens rearrangement in organic chemistry is an organic reaction converting quaternary ammonium salts and sulfonium salts to the corresponding amines or sulfides in presence of a strong base in a 1,2-rearrangement....
- Wagner–Meerwein rearrangement
- Westphalen–Lettré rearrangement
- Wolff rearrangementWolff rearrangementThe Wolff rearrangement is a rearrangement reaction converting a α-diazo-ketone into a ketene. This reaction was first reported by Ludwig Wolff in 1912....
1,3-Rearrangements
1,3-rearrangements take place over 3 carbon atoms. Examples:- the Fries rearrangementFries rearrangementThe Fries rearrangement, named for the German chemist Karl Theophil Fries, is a rearrangement reaction of a phenyl ester to a hydroxy aryl ketone by catalysis of Lewis acids.It involves migration of an acyl group of phenyl ester to benzene ring.- Mechanism:...
- a 1,3-alkyl shift of verbenoneVerbenoneVerbenone is a natural organic compound classified as a terpene which is found naturally in a variety of plants. The chemical has a pleasant characteristic odor. Besides being a natural constituent of plants, it and its analogs are insect pheromones...
to chrysanthenoneChrysanthenoneChrysanthenone is a terpene. It can be produced from its isomer verbenone in a photochemical rearrangement reaction.-External links:*...

