.gif)
Phase (matter)
Encyclopedia
In the physical science
s, a phase is a region of space (a thermodynamic system
), throughout which all physical properties of a material are essentially uniform. Examples of physical properties include density
, index of refraction
, and chemical composition. A simple description is that a phase is a region of material that is chemically uniform, physically distinct, and (often) mechanically separable. In a system consisting of ice and water in a glass jar, the ice cubes are one phase, the water is a second phase, and the humid air over the water is a third phase. The glass of the jar is another separate phase. (See State of Matter#Glass)
The term phase is sometimes used as a synonym for state of matter
. Also, the term phase is sometimes used to refer to a set of equilibrium states demarcated in terms of state variables such as pressure and temperature by a phase boundary
on a phase diagram
. Because phase boundaries relate to changes in the organization of matter, such as a change from liquid to solid or a more subtle change from one crystal structure to another, this latter usage is similar to the use of "phase" as a synonym for state of matter. However, the state of matter and phase diagram usages are not commensurate with the formal definition given above and the intended meaning must be determined in part from the context in which the term is used.

 Distinct phases may be described as different states of matter
Distinct phases may be described as different states of matter
such as gas
, liquid
, solid
, plasma
or Bose–Einstein condensate
. Useful mesophase
s between solid and liquid form other states of matter.
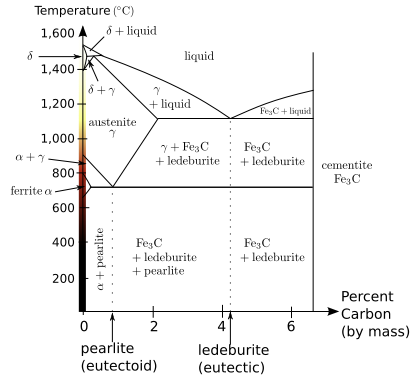
Distinct phases may also exist within a given state of matter. As shown in the diagram for iron alloys, several phases exist for both the solid and liquid states. Phases may also be differentiated based on solubility
as in polar (hydrophilic) or non-polar (hydrophobic). A mixture of water (a polar liquid) and oil (a non-polar liquid) will spontaneously separate into two phases. Water has a very low solubility
(is insoluble) in oil, and oil has a low solubility in water. Solubility is the maximum amount of a solute that can dissolve in a solvent before the solute ceases to dissolve and remains in a separate phase. A mixture can separate into more than two liquid phases and the concept of phase separation extends to solids, i.e., solids can form solid solution
s or crystallize into distinct crystal phases. Metal pairs that are mutually soluble can form alloy
s, whereas metal pair that are mutually insoluble cannot.
As many as eight immiscible liquid phases have been observed. Mutually immiscible liquid phases are formed from water (aqueous phase), hydrophobic organic solvents, perfluorocarbons (fluorous phase), silicones, several different metals, and also from molten phosphorus. Not all organic solvents are completely miscible, e.g. a mixture of ethylene glycol
and toluene
may separate into two distinct organic phases.
Phases do not need to macroscopically separate spontaneously. Emulsion
s and colloid
s are examples of immiscible phase pair combinations that do not physically separate.
Water in a closed jar with an air space over it forms a two phase system. Most of the water is in the liquid phase, where it is held by the mutual attraction of water molecules. Even at equilibrium molecules are constantly in motion and, once in a while, a molecule in the liquid phase gains enough kinetic energy to break away from the liquid phase and enter the gas phase. Likewise, every once in a while a vapor molecule collides with the liquid surface and condenses into the liquid. At equilibrium, evaporation and condensation processes exactly balance and there is no net change in the volume of either phase.
At room temperature and pressure, the water jar reaches equilibrium when the air over the water has a humidity of about 3%. This percentage increases as the temperature goes up. At 100 °C and atmospheric pressure, equilibrium is not reached until the air is 100% water. If the liquid is heated a little over 100 °C, the transition from liquid to gas will occur not only at the surface, but throughout the liquid volume: the water boils.
 For a given composition, only certain phases are possible at a given temperature
For a given composition, only certain phases are possible at a given temperature
and pressure. The number and type of phases that will form is hard to predict and is usually determined by experiment. The results of such experiments can be plotted in phase diagram
s.
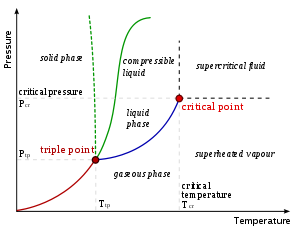
The phase diagram shown here is for a single component system. In this simple system, which phases that are possible depends only on pressure
and temperature
. The markings show points where two or more phases can co-exist in equilibrium. At temperatures and pressures away from the markings, there will be only one phase at equilibrium.
In the diagram, the blue line marking the boundary between liquid and gas does not continue indefinitely, but terminates at a point called the critical point
. As the temperature and pressure approach the critical point, the properties of the liquid and gas become progressively more similar. At the critical point, the liquid and gas become indistinguishable. Above the critical point, there are no longer separate liquid and gas phases: there is only a generic fluid phase referred to as a supercritical fluid
. In water, the critical point occurs at around 647 K
(374 °C or 705 °F) and 22.064 MPa.
An unusual feature of the water phase diagram is that the solid–liquid phase line (illustrated by the dotted green line) has a negative slope. For most substances, the slope is positive as exemplified by the dark green line. This unusual feature of water is related to ice having a lower density than liquid water. Increasing the pressure drives the water into the higher density phase, which causes melting.
Another interesting though not unusual feature of the phase diagram is the point where the solid–liquid phase line meets the liquid–gas phase line. The intersection is referred to as the triple point
. At the triple point, all three phases can coexist.
Experimentally, the phase lines are relatively easy to map due to the interdependence of temperature and pressure that develops when multiple phases forms. See Gibbs' phase rule
. Consider a test apparatus consisting of a closed and well insulated cylinder equipped with a piston. By charging the right amount of water and applying heat, the system can be brought to any point in the gas region of the phase diagram. If the piston is slowly lowered, the system will trace a curve of increasing temperature and pressure within the gas region of the phase diagram. At the point where liquid begins to condense, the direction of the temperature and pressure curve will abruptly change to trace along the phase line until all of the water has condensed.
. In mixtures, some components may preferentially move toward the interface. In terms of modeling, describing, or understanding the behavior of a particular system, it may be efficacious to treat the interfacial region as a separate phase.
is ordinarily found in the hexagonal form ice Ih
, but can also exist as the cubic ice Ic
, the rhombohedral
ice II
, and many other forms. Polymorphism
is the ability of a solid to exist in more than one crystal form. For pure chemical elements, polymorphism is known as allotropy
. For example, diamond
, graphite
, and fullerene
s are different allotropes of carbon
.
to another) it usually either takes up or releases energy. For example, when water evaporates, the kinetic energy expended as the evaporating molecules escape the attractive forces of the liquid is reflected in a decrease in temperature. The amount of energy required to induce the transition is more than the amount required to heat the water from room temperature to just short of boiling temperature, which is why evaporation is useful for cooling. See Enthalpy of vaporization. The reverse process, condensation, releases heat. The heat energy, or enthalpy, associated with a solid to liquid transition is the enthalpy of fusion
and that associated with a solid to gas transition is the enthalpy of sublimation
.
Physical science
Physical science is an encompassing term for the branches of natural science and science that study non-living systems, in contrast to the life sciences...
s, a phase is a region of space (a thermodynamic system
Thermodynamic system
A thermodynamic system is a precisely defined macroscopic region of the universe, often called a physical system, that is studied using the principles of thermodynamics....
), throughout which all physical properties of a material are essentially uniform. Examples of physical properties include density
Density
The mass density or density of a material is defined as its mass per unit volume. The symbol most often used for density is ρ . In some cases , density is also defined as its weight per unit volume; although, this quantity is more properly called specific weight...
, index of refraction
Refractive index
In optics the refractive index or index of refraction of a substance or medium is a measure of the speed of light in that medium. It is expressed as a ratio of the speed of light in vacuum relative to that in the considered medium....
, and chemical composition. A simple description is that a phase is a region of material that is chemically uniform, physically distinct, and (often) mechanically separable. In a system consisting of ice and water in a glass jar, the ice cubes are one phase, the water is a second phase, and the humid air over the water is a third phase. The glass of the jar is another separate phase. (See State of Matter#Glass)
The term phase is sometimes used as a synonym for state of matter
State of matter
States of matter are the distinct forms that different phases of matter take on. Solid, liquid and gas are the most common states of matter on Earth. However, much of the baryonic matter of the universe is in the form of hot plasma, both as rarefied interstellar medium and as dense...
. Also, the term phase is sometimes used to refer to a set of equilibrium states demarcated in terms of state variables such as pressure and temperature by a phase boundary
Phase boundary
The behavior of phase boundaries has been a developing subject of interest and an active research field in physics and mathematics for almost two centuries. One reason behind this is that phase boundaries naturally arise in many physical processes due to immiscibility of two or more substances with...
on a phase diagram
Phase diagram
A phase diagram in physical chemistry, engineering, mineralogy, and materials science is a type of chart used to show conditions at which thermodynamically distinct phases can occur at equilibrium...
. Because phase boundaries relate to changes in the organization of matter, such as a change from liquid to solid or a more subtle change from one crystal structure to another, this latter usage is similar to the use of "phase" as a synonym for state of matter. However, the state of matter and phase diagram usages are not commensurate with the formal definition given above and the intended meaning must be determined in part from the context in which the term is used.

Types of phases

State of matter
States of matter are the distinct forms that different phases of matter take on. Solid, liquid and gas are the most common states of matter on Earth. However, much of the baryonic matter of the universe is in the form of hot plasma, both as rarefied interstellar medium and as dense...
such as gas
Gas
Gas is one of the three classical states of matter . Near absolute zero, a substance exists as a solid. As heat is added to this substance it melts into a liquid at its melting point , boils into a gas at its boiling point, and if heated high enough would enter a plasma state in which the electrons...
, liquid
Liquid
Liquid is one of the three classical states of matter . Like a gas, a liquid is able to flow and take the shape of a container. Some liquids resist compression, while others can be compressed. Unlike a gas, a liquid does not disperse to fill every space of a container, and maintains a fairly...
, solid
Solid
Solid is one of the three classical states of matter . It is characterized by structural rigidity and resistance to changes of shape or volume. Unlike a liquid, a solid object does not flow to take on the shape of its container, nor does it expand to fill the entire volume available to it like a...
, plasma
Plasma (physics)
In physics and chemistry, plasma is a state of matter similar to gas in which a certain portion of the particles are ionized. Heating a gas may ionize its molecules or atoms , thus turning it into a plasma, which contains charged particles: positive ions and negative electrons or ions...
or Bose–Einstein condensate
Bose–Einstein condensate
A Bose–Einstein condensate is a state of matter of a dilute gas of weakly interacting bosons confined in an external potential and cooled to temperatures very near absolute zero . Under such conditions, a large fraction of the bosons occupy the lowest quantum state of the external potential, at...
. Useful mesophase
Mesophase
In physics, a mesophase is a state of matter intermediate between liquid and solid. Gelatin is a common example of a partially-ordered structure in a mesophase...
s between solid and liquid form other states of matter.
Distinct phases may also exist within a given state of matter. As shown in the diagram for iron alloys, several phases exist for both the solid and liquid states. Phases may also be differentiated based on solubility
Solubility
Solubility is the property of a solid, liquid, or gaseous chemical substance called solute to dissolve in a solid, liquid, or gaseous solvent to form a homogeneous solution of the solute in the solvent. The solubility of a substance fundamentally depends on the used solvent as well as on...
as in polar (hydrophilic) or non-polar (hydrophobic). A mixture of water (a polar liquid) and oil (a non-polar liquid) will spontaneously separate into two phases. Water has a very low solubility
Solubility
Solubility is the property of a solid, liquid, or gaseous chemical substance called solute to dissolve in a solid, liquid, or gaseous solvent to form a homogeneous solution of the solute in the solvent. The solubility of a substance fundamentally depends on the used solvent as well as on...
(is insoluble) in oil, and oil has a low solubility in water. Solubility is the maximum amount of a solute that can dissolve in a solvent before the solute ceases to dissolve and remains in a separate phase. A mixture can separate into more than two liquid phases and the concept of phase separation extends to solids, i.e., solids can form solid solution
Solid solution
A solid solution is a solid-state solution of one or more solutes in a solvent. Such a mixture is considered a solution rather than a compound when the crystal structure of the solvent remains unchanged by addition of the solutes, and when the mixture remains in a single homogeneous phase...
s or crystallize into distinct crystal phases. Metal pairs that are mutually soluble can form alloy
Alloy
An alloy is a mixture or metallic solid solution composed of two or more elements. Complete solid solution alloys give single solid phase microstructure, while partial solutions give two or more phases that may or may not be homogeneous in distribution, depending on thermal history...
s, whereas metal pair that are mutually insoluble cannot.
As many as eight immiscible liquid phases have been observed. Mutually immiscible liquid phases are formed from water (aqueous phase), hydrophobic organic solvents, perfluorocarbons (fluorous phase), silicones, several different metals, and also from molten phosphorus. Not all organic solvents are completely miscible, e.g. a mixture of ethylene glycol
Ethylene glycol
Ethylene glycol is an organic compound widely used as an automotive antifreeze and a precursor to polymers. In its pure form, it is an odorless, colorless, syrupy, sweet-tasting liquid...
and toluene
Toluene
Toluene, formerly known as toluol, is a clear, water-insoluble liquid with the typical smell of paint thinners. It is a mono-substituted benzene derivative, i.e., one in which a single hydrogen atom from the benzene molecule has been replaced by a univalent group, in this case CH3.It is an aromatic...
may separate into two distinct organic phases.
Phases do not need to macroscopically separate spontaneously. Emulsion
Emulsion
An emulsion is a mixture of two or more liquids that are normally immiscible . Emulsions are part of a more general class of two-phase systems of matter called colloids. Although the terms colloid and emulsion are sometimes used interchangeably, emulsion is used when both the dispersed and the...
s and colloid
Colloid
A colloid is a substance microscopically dispersed evenly throughout another substance.A colloidal system consists of two separate phases: a dispersed phase and a continuous phase . A colloidal system may be solid, liquid, or gaseous.Many familiar substances are colloids, as shown in the chart below...
s are examples of immiscible phase pair combinations that do not physically separate.
Phase equilibrium
Left to equilibration, many compositions will form a uniform single phase, but depending on the temperature and pressure even a single substance may separate into two or more distinct phases. Within each phase, the properties are uniform but between the two phases properties differ.Water in a closed jar with an air space over it forms a two phase system. Most of the water is in the liquid phase, where it is held by the mutual attraction of water molecules. Even at equilibrium molecules are constantly in motion and, once in a while, a molecule in the liquid phase gains enough kinetic energy to break away from the liquid phase and enter the gas phase. Likewise, every once in a while a vapor molecule collides with the liquid surface and condenses into the liquid. At equilibrium, evaporation and condensation processes exactly balance and there is no net change in the volume of either phase.
At room temperature and pressure, the water jar reaches equilibrium when the air over the water has a humidity of about 3%. This percentage increases as the temperature goes up. At 100 °C and atmospheric pressure, equilibrium is not reached until the air is 100% water. If the liquid is heated a little over 100 °C, the transition from liquid to gas will occur not only at the surface, but throughout the liquid volume: the water boils.
Number of phases

Temperature
Temperature is a physical property of matter that quantitatively expresses the common notions of hot and cold. Objects of low temperature are cold, while various degrees of higher temperatures are referred to as warm or hot...
and pressure. The number and type of phases that will form is hard to predict and is usually determined by experiment. The results of such experiments can be plotted in phase diagram
Phase diagram
A phase diagram in physical chemistry, engineering, mineralogy, and materials science is a type of chart used to show conditions at which thermodynamically distinct phases can occur at equilibrium...
s.
The phase diagram shown here is for a single component system. In this simple system, which phases that are possible depends only on pressure
Pressure
Pressure is the force per unit area applied in a direction perpendicular to the surface of an object. Gauge pressure is the pressure relative to the local atmospheric or ambient pressure.- Definition :...
and temperature
Temperature
Temperature is a physical property of matter that quantitatively expresses the common notions of hot and cold. Objects of low temperature are cold, while various degrees of higher temperatures are referred to as warm or hot...
. The markings show points where two or more phases can co-exist in equilibrium. At temperatures and pressures away from the markings, there will be only one phase at equilibrium.
In the diagram, the blue line marking the boundary between liquid and gas does not continue indefinitely, but terminates at a point called the critical point
Critical point (thermodynamics)
In physical chemistry, thermodynamics, chemistry and condensed matter physics, a critical point, also called a critical state, specifies the conditions at which a phase boundary ceases to exist...
. As the temperature and pressure approach the critical point, the properties of the liquid and gas become progressively more similar. At the critical point, the liquid and gas become indistinguishable. Above the critical point, there are no longer separate liquid and gas phases: there is only a generic fluid phase referred to as a supercritical fluid
Supercritical fluid
A supercritical fluid is any substance at a temperature and pressure above its critical point, where distinct liquid and gas phases do not exist. It can effuse through solids like a gas, and dissolve materials like a liquid...
. In water, the critical point occurs at around 647 K
Kelvin
The kelvin is a unit of measurement for temperature. It is one of the seven base units in the International System of Units and is assigned the unit symbol K. The Kelvin scale is an absolute, thermodynamic temperature scale using as its null point absolute zero, the temperature at which all...
(374 °C or 705 °F) and 22.064 MPa.
An unusual feature of the water phase diagram is that the solid–liquid phase line (illustrated by the dotted green line) has a negative slope. For most substances, the slope is positive as exemplified by the dark green line. This unusual feature of water is related to ice having a lower density than liquid water. Increasing the pressure drives the water into the higher density phase, which causes melting.
Another interesting though not unusual feature of the phase diagram is the point where the solid–liquid phase line meets the liquid–gas phase line. The intersection is referred to as the triple point
Triple point
In thermodynamics, the triple point of a substance is the temperature and pressure at which the three phases of that substance coexist in thermodynamic equilibrium...
. At the triple point, all three phases can coexist.
Experimentally, the phase lines are relatively easy to map due to the interdependence of temperature and pressure that develops when multiple phases forms. See Gibbs' phase rule
Gibbs' phase rule
Gibbs' phase rule was proposed by Josiah Willard Gibbs in the 1870s as the equalityF\;=\;C\;-\;P\;+\;2where P is the number of phases in thermodynamic equilibrium with each other and C is the number of components. Typical phases are solids, liquids and gases. A system involving one pure chemical...
. Consider a test apparatus consisting of a closed and well insulated cylinder equipped with a piston. By charging the right amount of water and applying heat, the system can be brought to any point in the gas region of the phase diagram. If the piston is slowly lowered, the system will trace a curve of increasing temperature and pressure within the gas region of the phase diagram. At the point where liquid begins to condense, the direction of the temperature and pressure curve will abruptly change to trace along the phase line until all of the water has condensed.
Interfacial phenomena
Between two phases in equilibrium there is a narrow region where the properties are not that of either phase. Although this region may be very thin, it can have significant and easily observable effects, such as causing a liquid to exhibit surface tensionSurface tension
Surface tension is a property of the surface of a liquid that allows it to resist an external force. It is revealed, for example, in floating of some objects on the surface of water, even though they are denser than water, and in the ability of some insects to run on the water surface...
. In mixtures, some components may preferentially move toward the interface. In terms of modeling, describing, or understanding the behavior of a particular system, it may be efficacious to treat the interfacial region as a separate phase.
Crystal phases
A single material may have several distinct solid states capable of forming separate phases. Water is a well known example of such a material. For example, water iceIce
Ice is water frozen into the solid state. Usually ice is the phase known as ice Ih, which is the most abundant of the varying solid phases on the Earth's surface. It can appear transparent or opaque bluish-white color, depending on the presence of impurities or air inclusions...
is ordinarily found in the hexagonal form ice Ih
Ice Ih
thumb|Photograph showing details of an ice cube under magnification. Ice Ih is the form of ice commonly seen on earth.Ice Ih is the hexagonal crystal form of ordinary ice, or frozen water. Virtually all ice in the biosphere is ice Ih, with the exception only of a small amount of ice Ic which is...
, but can also exist as the cubic ice Ic
Ice Ic
Ice Ic is a metastable cubic crystalline variant of ice. The oxygen atoms are arranged in a diamond structure. It is produced at temperatures between 130 and 220 K , and can exist up to 240 K, when it transforms into ice Ih. It may occasionally be present in the upper atmosphere.Ordinary water ice...
, the rhombohedral
Rhombohedral crystal system
In crystallography, the trigonal crystal system is one of the seven crystal systems, and the rhombohedral lattice system is one of the seven lattice systems...
ice II
Ice II
Ice II is a rhombohedral crystalline form of ice with highly ordered structure. It is formed from ice Ih by compressing it at temperature of 198 K at 300 MPa or by decompressing ice V. When heated it undergoes transformation to ice III....
, and many other forms. Polymorphism
Polymorphism (materials science)
Polymorphism in materials science is the ability of a solid material to exist in more than one form or crystal structure. Polymorphism can potentially be found in any crystalline material including polymers, minerals, and metals, and is related to allotropy, which refers to chemical elements...
is the ability of a solid to exist in more than one crystal form. For pure chemical elements, polymorphism is known as allotropy
Allotropy
Allotropy or allotropism is the property of some chemical elements to exist in two or more different forms, known as allotropes of these elements...
. For example, diamond
Diamond
In mineralogy, diamond is an allotrope of carbon, where the carbon atoms are arranged in a variation of the face-centered cubic crystal structure called a diamond lattice. Diamond is less stable than graphite, but the conversion rate from diamond to graphite is negligible at ambient conditions...
, graphite
Graphite
The mineral graphite is one of the allotropes of carbon. It was named by Abraham Gottlob Werner in 1789 from the Ancient Greek γράφω , "to draw/write", for its use in pencils, where it is commonly called lead . Unlike diamond , graphite is an electrical conductor, a semimetal...
, and fullerene
Fullerene
A fullerene is any molecule composed entirely of carbon, in the form of a hollow sphere, ellipsoid, or tube. Spherical fullerenes are also called buckyballs, and they resemble the balls used in association football. Cylindrical ones are called carbon nanotubes or buckytubes...
s are different allotropes of carbon
Carbon
Carbon is the chemical element with symbol C and atomic number 6. As a member of group 14 on the periodic table, it is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds...
.
Phase transitions
When a substance undergoes a phase transition (changes from one state of matterState of matter
States of matter are the distinct forms that different phases of matter take on. Solid, liquid and gas are the most common states of matter on Earth. However, much of the baryonic matter of the universe is in the form of hot plasma, both as rarefied interstellar medium and as dense...
to another) it usually either takes up or releases energy. For example, when water evaporates, the kinetic energy expended as the evaporating molecules escape the attractive forces of the liquid is reflected in a decrease in temperature. The amount of energy required to induce the transition is more than the amount required to heat the water from room temperature to just short of boiling temperature, which is why evaporation is useful for cooling. See Enthalpy of vaporization. The reverse process, condensation, releases heat. The heat energy, or enthalpy, associated with a solid to liquid transition is the enthalpy of fusion
Enthalpy of fusion
The enthalpy of fusion is the change in enthalpy resulting from heating one mole of a substance to change its state from a solid to a liquid. The temperature at which this occurs is the melting point....
and that associated with a solid to gas transition is the enthalpy of sublimation
Enthalpy of sublimation
The enthalpy of sublimation, or heat of sublimation, is defined as the heat required to sublime one mole of the substance at a given combination of temperature and pressure, usually standard temperature and pressure...
.
External links
- French physicists find a solution that reversibly solidifies with a rise in temperature – α-cyclodextrinCyclodextrinCyclodextrins are a family of compounds made up of sugar molecules bound together in a ring ....
, waterWaterWater is a chemical substance with the chemical formula H2O. A water molecule contains one oxygen and two hydrogen atoms connected by covalent bonds. Water is a liquid at ambient conditions, but it often co-exists on Earth with its solid state, ice, and gaseous state . Water also exists in a...
, and 4-methylpyridine

