
Hydrophobe
Encyclopedia


Chemistry
Chemistry is the science of matter, especially its chemical reactions, but also its composition, structure and properties. Chemistry is concerned with atoms and their interactions with other atoms, and particularly with the properties of chemical bonds....
, hydrophobicity (from the combining form of water in Attic Greek
Attic Greek
Attic Greek is the prestige dialect of Ancient Greek that was spoken in Attica, which includes Athens. Of the ancient dialects, it is the most similar to later Greek, and is the standard form of the language studied in courses of "Ancient Greek". It is sometimes included in Ionic.- Origin and range...
hydro- and for fear phobos) is the physical property of a molecule
Molecule
A molecule is an electrically neutral group of at least two atoms held together by covalent chemical bonds. Molecules are distinguished from ions by their electrical charge...
(known as a hydrophobe) that is repelled from a mass of water
Water
Water is a chemical substance with the chemical formula H2O. A water molecule contains one oxygen and two hydrogen atoms connected by covalent bonds. Water is a liquid at ambient conditions, but it often co-exists on Earth with its solid state, ice, and gaseous state . Water also exists in a...
.
Hydrophobic molecules tend to be non-polar and thus prefer other neutral molecules and non-polar solvent
Solvent
A solvent is a liquid, solid, or gas that dissolves another solid, liquid, or gaseous solute, resulting in a solution that is soluble in a certain volume of solvent at a specified temperature...
s. Hydrophobic molecules in water often cluster together, forming micelles. Water on hydrophobic surfaces will exhibit a high contact angle
Contact angle
The contact angle is the angle at which a liquid/vapor interface meets a solid surface. The contact angle is specific for any given system and is determined by the interactions across the three interfaces. Most often the concept is illustrated with a small liquid droplet resting on a flat...
.
Examples of hydrophobic molecules include the alkanes, oil
Oil
An oil is any substance that is liquid at ambient temperatures and does not mix with water but may mix with other oils and organic solvents. This general definition includes vegetable oils, volatile essential oils, petrochemical oils, and synthetic oils....
s, fat
Fat
Fats consist of a wide group of compounds that are generally soluble in organic solvents and generally insoluble in water. Chemically, fats are triglycerides, triesters of glycerol and any of several fatty acids. Fats may be either solid or liquid at room temperature, depending on their structure...
s, and greasy substances in general. Hydrophobic materials are used for oil removal from water, the management of oil spills, and chemical separation processes to remove non-polar from polar compounds.
Hydrophobic is often used interchangeably with lipophilic
Lipophilic
Lipophilicity, , refers to the ability of a chemical compound to dissolve in fats, oils, lipids, and non-polar solvents such as hexane or toluene. These non-polar solvents are themselves lipophilic — the axiom that like dissolves like generally holds true...
, "fat loving." However, the two terms are not synonymous. While hydrophobic substances are usually lipophilic, there are exceptions—such as the silicones and fluorocarbon
Fluorocarbon
Fluorocarbons, sometimes referred to as perfluorocarbons or PFCs, are organofluorine compounds that contain only carbon and fluorine bonded together in strong carbon–fluorine bonds. Fluoroalkanes that contain only single bonds are more chemically and thermally stable than alkanes...
s.
Chemical background
According to thermodynamicsThermodynamics
Thermodynamics is a physical science that studies the effects on material bodies, and on radiation in regions of space, of transfer of heat and of work done on or by the bodies or radiation...
, matter seeks to be in a low-energy state, and bonding reduces chemical energy. Water is electrically polarized, and is able to form hydrogen bond
Hydrogen bond
A hydrogen bond is the attractive interaction of a hydrogen atom with an electronegative atom, such as nitrogen, oxygen or fluorine, that comes from another molecule or chemical group. The hydrogen must be covalently bonded to another electronegative atom to create the bond...
s internally, which gives it many of its unique physical properties. But, since hydrophobes are not electrically polarized, and because they are unable to form hydrogen bonds, water repels hydrophobes, in favour of bonding with itself. It is this effect that causes the hydrophobic interaction
Hydrophobic effect
The hydrophobic effect is the observed tendency of nonpolar substances to aggregate in aqueous solution and exclude water molecules. The name, literally meaning "water-fearing," describes the segregation and apparent repulsion between water and nonpolar substances...
—which in itself is misleadingly named as the energetic force comes from the hydrophilic molecules. Thus the two immiscible phases (hydrophilic vs. hydrophobic) will change so that their corresponding interfacial area will be minimal. This effect can be visualized in the phenomenon called phase
Phase (matter)
In the physical sciences, a phase is a region of space , throughout which all physical properties of a material are essentially uniform. Examples of physical properties include density, index of refraction, and chemical composition...
separation.
Superhydrophobicity

Contact angle
The contact angle is the angle at which a liquid/vapor interface meets a solid surface. The contact angle is specific for any given system and is determined by the interactions across the three interfaces. Most often the concept is illustrated with a small liquid droplet resting on a flat...
s of a water droplet exceeds 150° and the roll-off angle is less than 10°. This is referred to as the Lotus effect
Lotus effect
The lotus effect refers to the very high water repellence exhibited by the leaves of the lotus flower ....
.
Theory
In 1805, Thomas Young defined the contact angle θ by analyzing the forces acting on a fluid droplet resting on a solid surface surrounded by a gas.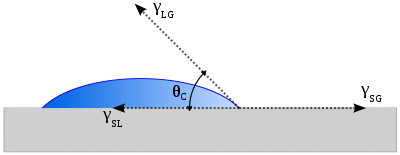
where
-
 = Interfacial tension between the solid and gas
= Interfacial tension between the solid and gas -
 = Interfacial tension between the solid and liquid
= Interfacial tension between the solid and liquid -
 = Interfacial tension between the liquid and gas
= Interfacial tension between the liquid and gas
θ can be measured using a contact angle goniometer
Goniometer
A goniometer is an instrument that either measures an angle or allows an object to be rotated to a precise angular position. The term goniometry is derived from two Greek words, gōnia, meaning angle, and metron, meaning measure....
.
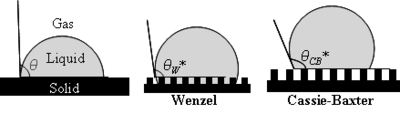
Wenzel determined that when the liquid is in intimate contact with a microstructured surface, θ will change to θW*
where r is the ratio of the actual area to the projected area. Wenzel's equation shows that microstructuring a surface amplifies the natural tendency of the surface. A hydrophobic surface (one that has an original contact angle greater than 90°) becomes more hydrophobic when microstructured – its new contact angle becomes greater than the original. However, a hydrophilic surface (one that has an original contact angle less than 90°) becomes more hydrophilic when microstructured – its new contact angle becomes less than the original.
Cassie and Baxter found that if the liquid is suspended on the tops of microstructures, θ will change to θCB*:
where φ is the area fraction of the solid that touches the liquid. Liquid in the Cassie–Baxter state is more mobile than in the Wenzel state.
We can predict whether the Wenzel or Cassie–Baxter state should exist by calculating the new contact angle with both equations. By a minimization of free energy argument, the relation that predicted the smaller new contact angle is the state most likely to exist. Stated mathematically, for the Cassie–Baxter state to exist, the following inequality must be true.
A recent alternative criterion for the Cassie–Baxter state asserts that the Cassie–Baxter state exists when the following 2 criteria are met: 1) Contact line forces overcome body forces of unsupported droplet weight and 2) The microstructures are tall enough to prevent the liquid that bridges microstructures from touching the base of the microstructures.
A new criterion for the switch between Wenzel and Cassie-Baxter states has been developed recently based on surface roughness and surface energy. The criterion focuses on the air-trapping capability under liquid droplets on rough surfaces, which could tell whether Wenzel's model or Cassie-Baster's model should be used for certain combination of surface roughness and energy.
Contact angle is a measure of static hydrophobicity, and contact angle hysteresis and slide angle are dynamic measures. Contact angle hysteresis is a phenomenon that characterizes surface heterogeneity. When a pipette injects a liquid onto a solid, the liquid will form some contact angle. As the pipette injects more liquid, the droplet will increase in volume, the contact angle will increase, but its three phase boundary will remain stationary until it suddenly advances outward. The contact angle the droplet had immediately before advancing outward is termed the advancing contact angle. The receding contact angle is now measured by pumping the liquid back out of the droplet. The droplet will decrease in volume, the contact angle will decrease, but its three phase boundary will remain stationary until it suddenly recedes inward. The contact angle the droplet had immediately before receding inward is termed the receding contact angle. The difference between advancing and receding contact angles is termed contact angle hysteresis and can be used to characterize surface heterogeneity, roughness, and mobility. Surfaces that are not homogeneous will have domains which impede motion of the contact line. The slide angle is another dynamic measure of hydrophobicity and is measured by depositing a droplet on a surface and tilting the surface until the droplet begins to slide. Liquids in the Cassie–Baxter state generally exhibit lower slide angles and contact angle hysteresis than those in the Wenzel state.
Research and development
The self-cleaning property of superhydrophobic micro-nanostructuredNanotechnology
Nanotechnology is the study of manipulating matter on an atomic and molecular scale. Generally, nanotechnology deals with developing materials, devices, or other structures possessing at least one dimension sized from 1 to 100 nanometres...
surfaces was reported in 1977, and perfluoroalkyl and perfluoropolyether superhydrophobic materials were developed in 1986 for handling chemical and biological fluids. Other biotechnical applications have emerged since the 1990s.
In recent research, superhydrophobicity has been reported by allowing alkylketene dimer (AKD) to solidify into a nanostructured fractal surface. Many papers have since presented fabrication methods for producing superhydrophobic surfaces including particle deposition, sol-gel techniques, plasma treatments, vapor deposition, and casting techniques. Current opportunity for research impact lies mainly in fundamental research and practical manufacturing. Debates have recently emerged concerning the applicability of the Wenzel and Cassie–Baxter models. In an experiment designed to challenge the surface energy perspective of the Wenzel and Cassie–Baxter model and promote a contact line perspective, water drops were placed on a smooth hydrophobic spot in a rough hydrophobic field, a rough hydrophobic spot in a smooth hydrophobic field, and a hydrophilic spot in a hydrophobic field. Experiments showed that the surface chemistry and geometry at the contact line affected the contact angle and contact angle hysteresis, but the surface area inside the contact line had no effect. An argument that increased jaggedness in the contact line enhances droplet mobility has also been proposed.
Many very hydrophobic materials found in nature rely on Cassie's law
Cassie's law
Cassie's law describes the effective contact angle θc for a liquid on a composite surface . The law explains how simply roughing up a surface increases the apparent surface angle...
and are biphasic
Phase (matter)
In the physical sciences, a phase is a region of space , throughout which all physical properties of a material are essentially uniform. Examples of physical properties include density, index of refraction, and chemical composition...
on the submicrometer level with one component air. The Lotus effect
Lotus effect
The lotus effect refers to the very high water repellence exhibited by the leaves of the lotus flower ....
is based on this principle. Inspired by it, a lot of functional superhydrophobic surfaces were prepared.
An example of a biomimetic
Bionics
Bionics is the application of biological methods and systems found in nature to the study and design of engineering systems and modern technology.The word bionic was coined by Jack E...
superhydrophobic material in nanotechnology
Nanotechnology
Nanotechnology is the study of manipulating matter on an atomic and molecular scale. Generally, nanotechnology deals with developing materials, devices, or other structures possessing at least one dimension sized from 1 to 100 nanometres...
is nanopin film
Nanopin film
Nanopin film is an experimental material in nanotechnology developed in 2005 with unusual superhydrophobic properties . A droplet of water makes contact with the surface of this film and forms an almost perfect sphere with a contact angle of 178°. The film is able to do this because it is covered...
. In one study a vanadium pentoxide surface is presented that can switch reversibly between superhydrophobicity and superhydrophilicity under the influence of UV radiation. According to the study any surface can be modified to this effect by application of a suspension
Suspension (chemistry)
In chemistry, a suspension is a heterogeneous fluid containing solid particles that are sufficiently large for sedimentation. Usually they must be larger than 1 micrometer. The internal phase is dispersed throughout the external phase through mechanical agitation, with the use of certain...
of rose-like V2O5 particles for instance with an inkjet printer
Inkjet printer
An inkjet printer is a type of computer printer that creates a digital image by propelling droplets of ink onto paper. Inkjet printers are the most commonly used type of printer and range from small inexpensive consumer models to very large professional machines that can cost up to thousands of...
. Once again hydrophobicity is induced by interlaminar air pockets (separated by 2.1 nm distances). The UV effect is also explained. UV light creates electron-hole pairs, with the holes reacting with lattice oxygen creating surface oxygen vacancies while the electrons reduce V5+ to V3+. The oxygen vacancies are met by water and this water absorbency by the vanadium surface makes it hydrophilic. By extended storage in the dark, water is replaced by oxygen and hydrophilicity is once again lost.
Potential applications
Active recent research on superhydrophobic materials might eventually lead to industrial applications. For example, a simple routine of coating cotton fabric with silica or titaniaTitanium dioxide
Titanium dioxide, also known as titanium oxide or titania, is the naturally occurring oxide of titanium, chemical formula . When used as a pigment, it is called titanium white, Pigment White 6, or CI 77891. Generally it comes in two different forms, rutile and anatase. It has a wide range of...
particles by sol-gel technique has been reported, which protects the fabric from UV light and makes it superhydrophobic. Also, an efficient routine has been reported for making polyethylene
Polyethylene
Polyethylene or polythene is the most widely used plastic, with an annual production of approximately 80 million metric tons...
superhydrophobic and thus self-cleaning—99% of dirt adsorbed on such surface is easily washed away. Patterned superhydrophobic surfaces also have the promises for the lab-on-a-chip, microfluidic devices and can drastically improve the surface based bioanalysis.
See also
- Amphiphile
- Froth flotationFroth flotationFroth flotation is a process for selectively separating hydrophobic materials from hydrophilic. This is used in several processing industries...
- Hydrophilic
- Hydrophobic effectHydrophobic effectThe hydrophobic effect is the observed tendency of nonpolar substances to aggregate in aqueous solution and exclude water molecules. The name, literally meaning "water-fearing," describes the segregation and apparent repulsion between water and nonpolar substances...
- Hydrophobicity scalesHydrophobicity scalesHydrophobicity scales are ranking lists for the relative hydrophobicity of amino acid residues. In general, the more positive the value, the more hydrophobic are the amino acids located in that region of the protein...





