
Sulfone
Encyclopedia
Chemical compound
A chemical compound is a pure chemical substance consisting of two or more different chemical elements that can be separated into simpler substances by chemical reactions. Chemical compounds have a unique and defined chemical structure; they consist of a fixed ratio of atoms that are held together...
containing a sulfonyl
Sulfonyl
A sulfonyl group can refer either to a functional group found primarily in sulfones or to a substituent obtained from a sulfonic acid by the removal of the hydroxyl group similarly to acyl groups...
functional group
Functional group
In organic chemistry, functional groups are specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules. The same functional group will undergo the same or similar chemical reaction regardless of the size of the molecule it is a part of...
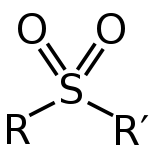
attached to two carbon atoms. The central hexavalent
Valence (chemistry)
In chemistry, valence, also known as valency or valence number, is a measure of the number of bonds formed by an atom of a given element. "Valence" can be defined as the number of valence bonds...
sulfur
Sulfur
Sulfur or sulphur is the chemical element with atomic number 16. In the periodic table it is represented by the symbol S. It is an abundant, multivalent non-metal. Under normal conditions, sulfur atoms form cyclic octatomic molecules with chemical formula S8. Elemental sulfur is a bright yellow...
atom is double bond
Double bond
A double bond in chemistry is a chemical bond between two chemical elements involving four bonding electrons instead of the usual two. The most common double bond, that between two carbon atoms, can be found in alkenes. Many types of double bonds between two different elements exist, for example in...
ed to each of two oxygen
Oxygen
Oxygen is the element with atomic number 8 and represented by the symbol O. Its name derives from the Greek roots ὀξύς and -γενής , because at the time of naming, it was mistakenly thought that all acids required oxygen in their composition...
atoms and has a single bond to each of two carbon atoms, usually in two separate hydrocarbon substituents.
IUPAC name and chemistry
The use of the long-standing alternative spelling sulphone is discouraged by IUPAC; it is definitely undesirable to have two spellings in simultaneous common use, and it was agreed to discontinue the ph spelling as the more archaic.The general structural formula
Structural formula
The structural formula of a chemical compound is a graphical representation of the molecular structure, showing how the atoms are arranged. The chemical bonding within the molecule is also shown, either explicitly or implicitly...
is R-S(=O)(=O)-R' where R and R' are the organic groups. Sulfide
Sulfide
A sulfide is an anion of sulfur in its lowest oxidation state of 2-. Sulfide is also a slightly archaic term for thioethers, a common type of organosulfur compound that are well known for their bad odors.- Properties :...
s are often the starting materials for sulfones by organic oxidation through the intermediate formation of sulfoxide
Sulfoxide
A sulfoxide is a chemical compound containing a sulfinyl functional group attached to two carbon atoms. Sulfoxides can be considered as oxidized sulfides...
s. For example dimethyl sulfide
Dimethyl sulfide
Dimethyl sulfide or methylthiomethane is an organosulfur compound with the formula 2S. Dimethyl sulfide is a water-insoluble flammable liquid that boils at and has a characteristic disagreeable odor. It is a component of the smell produced from cooking of certain vegetables, notably maize,...
is oxidized to dimethyl sulfoxide
Dimethyl sulfoxide
Dimethyl sulfoxide is an organosulfur compound with the formula 2SO. This colorless liquid is an important polar aprotic solvent that dissolves both polar and nonpolar compounds and is miscible in a wide range of organic solvents as well as water...
and then to dimethyl sulfone
Methylsulfonylmethane
Methylsulfonylmethane is an organosulfur compound with the formula 2SO2. It is also known by several other names including DMSO2, methyl sulfone, and dimethyl sulfone. This colorless solid features the sulfonyl functional group and is considered relatively inert chemically...
.
In the Ramberg-Bäcklund Reaction
Ramberg-Bäcklund reaction
The Ramberg-Bäcklund Reaction is an organic reaction converting an α-halo sulfone into an alkene in presence of a base with extrusion of sulfur dioxide. The reaction is named after the two Swedish chemists Ludwig Ramberg and Birger Bäcklund. The carbanion formed by deprotonation gives an unstable...
and the Julia olefination
Julia olefination
The Julia olefination is the chemical reaction of phenyl sulfones with aldehydes to give alkenes after alcohol functionalization and reductive elimination using sodium amalgam or SmI2...
sulfones are converted to alkene
Alkene
In organic chemistry, an alkene, olefin, or olefine is an unsaturated chemical compound containing at least one carbon-to-carbon double bond...
s through the elimination of sulfur dioxide
Sulfur dioxide
Sulfur dioxide is the chemical compound with the formula . It is released by volcanoes and in various industrial processes. Since coal and petroleum often contain sulfur compounds, their combustion generates sulfur dioxide unless the sulfur compounds are removed before burning the fuel...
.
Polymers
In recent decades sulfone polymers have gained prominence in the field of engineering plastics. Various polymers are of importance because of properties such as their high strength and their resistance to oxidation, corrosion, high temperatures, and creep under stress. For example, some are valuable as replacements for copper in domestic hot water plumbing.Pharmacology
Examples of sulfones in pharmacology include dapsoneDapsone
Dapsone is a medication most commonly used in combination with rifampicin and clofazimine as multidrug therapy for the treatment of Mycobacterium leprae infections . It is also second-line treatment for prophylaxis against Pneumocystis pneumonia caused by Pneumocystis jiroveci Dapsone...
, a drug formerly used as an antibiotic
Antibiotic
An antibacterial is a compound or substance that kills or slows down the growth of bacteria.The term is often used synonymously with the term antibiotic; today, however, with increased knowledge of the causative agents of various infectious diseases, antibiotic has come to denote a broader range of...
to treat leprosy
Leprosy
Leprosy or Hansen's disease is a chronic disease caused by the bacteria Mycobacterium leprae and Mycobacterium lepromatosis. Named after physician Gerhard Armauer Hansen, leprosy is primarily a granulomatous disease of the peripheral nerves and mucosa of the upper respiratory tract; skin lesions...
, dermatitis herpetiformis
Dermatitis herpetiformis
Dermatitis herpetiformis , or Duhring's disease,Freedberg, et al. . Fitzpatrick's Dermatology in General Medicine. . McGraw-Hill. ISBN 0-07-138076-0. is a chronic blistering skin condition, characterised by blisters filled with a watery fluid...
, tuberculosis
Tuberculosis
Tuberculosis, MTB, or TB is a common, and in many cases lethal, infectious disease caused by various strains of mycobacteria, usually Mycobacterium tuberculosis. Tuberculosis usually attacks the lungs but can also affect other parts of the body...
, or pneumocystis pneumonia
Pneumocystis pneumonia
Pneumocystis pneumonia or pneumocystosis is a form of pneumonia, caused by the yeast-like fungus Pneumocystis jirovecii...
(PCP). Several of its derivatives, such as promin
Promin
Promin, or sodium glucosulfone is a sulfone drug that was investigated for the treatment of malaria, tuberculosis and leprosy. It is broken down in the body to dapsone, which is the therapeutic form.-History:...
, have similarly been studied or actually been applied in medicine, but in general sulfones are of far less prominence in pharmacology than say, the sulfonamides
Sulfonamide (medicine)
Sulfonamide or sulphonamide is the basis of several groups of drugs. The original antibacterial sulfonamides are synthetic antimicrobial agents that contain the sulfonamide group. Some sulfonamides are also devoid of antibacterial activity, e.g., the anticonvulsant sultiame...
.
In the literature, particularly lay literature, sulfones commonly are confused with sulfonamides, though the latter have one carbon and one nitrogen atom attached to the sulfur atom, instead of two carbon atoms. The pharmacological mechanisms accordingly differ from that of the sulfonamides. However, in practice one commonly sees frequent references to dapsone and promin as sulfonamides. Probably this is partly because few pharmaceuticals are in fact sulfones.

