
Kosmotropic
Encyclopedia
Solutes
are defined as kosmotropic if they contribute to the stability and structure of water-water interactions. Kosmotropes cause water molecules to favorably interact, which also stabilizes intermolecular interactions in macromolecules such as proteins.
, phosphate, magnesium
(2+), lithium
(1+), zinc
(2+) and aluminium
(+3). Large ions or ions with low charge density (such as bromide
, iodide
, potassium
(1+), caesium
(1+)) instead act as chaotropes
.
A scale can be established if one refers to the Hofmeister
series or looks up the free energy
of hydration
(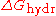 ) of the salts. The more negative
) of the salts. The more negative 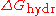 , the more kosmotropic the salt.
, the more kosmotropic the salt.
or urea are used to solubilise all proteins.
and glucose
as well as proline
and tert-butanol
are kosmotropes.
Solution
In chemistry, a solution is a homogeneous mixture composed of only one phase. In such a mixture, a solute is dissolved in another substance, known as a solvent. The solvent does the dissolving.- Types of solutions :...
are defined as kosmotropic if they contribute to the stability and structure of water-water interactions. Kosmotropes cause water molecules to favorably interact, which also stabilizes intermolecular interactions in macromolecules such as proteins.
Ionic Kosmotropes
Ionic kosmotropes tend to be small or have high charge density. Some ionic kosmotropes are sulfateSulfate
In inorganic chemistry, a sulfate is a salt of sulfuric acid.-Chemical properties:...
, phosphate, magnesium
Magnesium
Magnesium is a chemical element with the symbol Mg, atomic number 12, and common oxidation number +2. It is an alkaline earth metal and the eighth most abundant element in the Earth's crust and ninth in the known universe as a whole...
(2+), lithium
Lithium
Lithium is a soft, silver-white metal that belongs to the alkali metal group of chemical elements. It is represented by the symbol Li, and it has the atomic number 3. Under standard conditions it is the lightest metal and the least dense solid element. Like all alkali metals, lithium is highly...
(1+), zinc
Zinc
Zinc , or spelter , is a metallic chemical element; it has the symbol Zn and atomic number 30. It is the first element in group 12 of the periodic table. Zinc is, in some respects, chemically similar to magnesium, because its ion is of similar size and its only common oxidation state is +2...
(2+) and aluminium
Aluminium
Aluminium or aluminum is a silvery white member of the boron group of chemical elements. It has the symbol Al, and its atomic number is 13. It is not soluble in water under normal circumstances....
(+3). Large ions or ions with low charge density (such as bromide
Bromide
A bromide is a chemical compound containing bromide ion, that is bromine atom with effective charge of −1. The class name can include ionic compounds such as caesium bromide or covalent compounds such as sulfur dibromide.-Natural occurrence:...
, iodide
Iodide
An iodide ion is the ion I−. Compounds with iodine in formal oxidation state −1 are called iodides. This page is for the iodide ion and its salts. For information on organoiodides, see organohalides. In everyday life, iodide is most commonly encountered as a component of iodized salt,...
, potassium
Potassium
Potassium is the chemical element with the symbol K and atomic number 19. Elemental potassium is a soft silvery-white alkali metal that oxidizes rapidly in air and is very reactive with water, generating sufficient heat to ignite the hydrogen emitted in the reaction.Potassium and sodium are...
(1+), caesium
Caesium
Caesium or cesium is the chemical element with the symbol Cs and atomic number 55. It is a soft, silvery-gold alkali metal with a melting point of 28 °C , which makes it one of only five elemental metals that are liquid at room temperature...
(1+)) instead act as chaotropes
Chaotropic agent
A denaturating agent is a substance which disrupts the three dimensional structure in macromolecules such as proteins, DNA, or RNA and denatures them. A denaturating agent is a chaotropic agent, but chaotropic agents aren't necessarily denaturating agents...
.
A scale can be established if one refers to the Hofmeister
Franz Hofmeister
Franz Hofmeister was an early protein scientist, and is famous for his studies of salts that influence the solubility and conformational stability of proteins...
series or looks up the free energy
Gibbs free energy
In thermodynamics, the Gibbs free energy is a thermodynamic potential that measures the "useful" or process-initiating work obtainable from a thermodynamic system at a constant temperature and pressure...
of hydration
Hydration reaction
In organic chemistry, a hydration reaction is a chemical reaction in which a hydroxyl group and a hydrogen cation are added to the two carbon atoms bonded together in the carbon-carbon double bond which makes up an alkene functional group. The reaction usually runs in a strong acidic, aqueous...
(
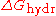 ) of the salts. The more negative
) of the salts. The more negative 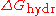 , the more kosmotropic the salt.
, the more kosmotropic the salt.Applications
Ammonium sulphate is the traditional kosmotropic salt for the salting out of protein from an aqueous solution. The chaotropic agents like Guanidinium chlorideGuanidinium chloride
Guanidinium chloride or guanidine hydrochloride, usually abbreviated GdmCl and sometimes GndCl or GuHCl, is the hydrochloride salt of guanidine.-Use in protein denaturation:...
or urea are used to solubilise all proteins.
Nonionic Kosmotropes
Nonionic kosmotropes have no net charge but are very soluble and become very hydrated. Carbohydrates such as trehaloseTrehalose
Trehalose, also known as mycose or tremalose, is a natural alpha-linked disaccharide formed by an α,α-1,1-glucoside bond between two α-glucose units. In 1832, H.A.L. Wiggers discovered trehalose in an ergot of rye, and in 1859 Marcellin Berthelot isolated it from trehala manna, a substance made...
and glucose
Glucose
Glucose is a simple sugar and an important carbohydrate in biology. Cells use it as the primary source of energy and a metabolic intermediate...
as well as proline
Proline
Proline is an α-amino acid, one of the twenty DNA-encoded amino acids. Its codons are CCU, CCC, CCA, and CCG. It is not an essential amino acid, which means that the human body can synthesize it. It is unique among the 20 protein-forming amino acids in that the α-amino group is secondary...
and tert-butanol
Tert-Butanol
tert-Butanol, or 2-methyl-2-propanol, is the simplest tertiary alcohol. It is one of the four isomers of butanol. tert-Butanol is a clear liquid with a camphor-like odor. It is very soluble in water and miscible with ethanol and diethyl ether...
are kosmotropes.
See also
- Chaotropic agentChaotropic agentA denaturating agent is a substance which disrupts the three dimensional structure in macromolecules such as proteins, DNA, or RNA and denatures them. A denaturating agent is a chaotropic agent, but chaotropic agents aren't necessarily denaturating agents...
and Guanidinium chlorideGuanidinium chlorideGuanidinium chloride or guanidine hydrochloride, usually abbreviated GdmCl and sometimes GndCl or GuHCl, is the hydrochloride salt of guanidine.-Use in protein denaturation:... - Protein precipitationProtein precipitationPrecipitation is widely used in downstream processing of biological products, such as proteins. This unit operation serves to concentrate and fractionate the target product from various contaminants. For example, in the biotechnology industry protein precipitation is used to eliminate contaminants...
, on aluminium sulphate "salting out"
External links
- Kosmotropes and Chaotropes (from Water Structure and Behavior)
- 'Zink sulphate' C. Polson et al., "Optimization of protein precipitation based upon effectiveness of protein removal and ionization effect in liquid chromatography–tandem mass spectrometry," Journal of Chromatography B, Volume 785 (263:275 )

