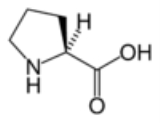
Proline
Encyclopedia
Proline is an α-amino acid
, one of the twenty DNA
-encoded amino acids. Its codons are CCU, CCC, CCA, and CCG. It is not an essential amino acid
, which means that the human body can synthesize it. It is unique among the 20 protein-forming amino acids in that the α-amino group is secondary. The more common L form has S stereochemistry.
derived from the amino acid L-glutamate and its immediate precursor is the imino acid
(S)-1-pyrroline-5-carboxylate
(P5C). Enzymes involved in a typical biosynthesis include:
at approximately −75°, giving proline an exceptional conformational rigidity compared to other amino acids. Hence, proline loses less conformational entropy
upon folding, which may account for its higher prevalence in the proteins of thermophilic
organisms. Proline acts as a structural disruptor in the middle of regular secondary structure
elements such as alpha helices
and beta sheet
s; however, proline is commonly found as the first residue of an alpha helix
and also in the edge strands of beta sheet
s. Proline is also commonly found in turns
, which may account for the curious fact that proline is usually solvent-exposed, despite having a completely aliphatic side chain. Because proline lacks a hydrogen on the amide group, it cannot act as a hydrogen bond
donor, only as a hydrogen bond
acceptor.
The distinct side chain/amine group interactions allow proline to aid in the formation of beta turns and also explains why it is not apparent in alpha helices...
 Multiple prolines and/or hydroxyproline
Multiple prolines and/or hydroxyproline
s in a row can create a polyproline helix
, the predominant secondary structure
in collagen
. The hydroxylation
of proline by prolyl hydroxylase
(or other additions of electron-withdrawing substituents such as fluorine
) increases the conformational stability of collagen
significantly. Hence, the hydroxylation of proline is a critical biochemical process for maintaining the connective tissue
of higher organisms. Severe diseases such as scurvy
can result from defects in this hydroxylation, e.g., mutations in the enzyme prolyl hydroxylase
or lack of the necessary ascorbate (vitamin C)
cofactor.
Sequences of proline and 2-aminoisobutyric acid
(Aib) also form a helical turn structure.
In 2006, scientists at ASU
discovered that solutions of TiO2
illuminated with ultraviolet
radiation can serve as an extremely cost-effective and accurate protein cleavage catalyst. The TiO2 catalyst preferentially and rapidly cleaves protein at sites where proline is present, while taking much longer to degrade the protein from its endpoints.
Peptide bond formation with incoming Pro-tRNAPro is considerably slower than with any other tRNAs, which is a general feature of N-alkylamino acids.
s to proline, and to other N-substituted amino acids (such as sarcosine
), are able to populate both the cis and trans isomers. Most peptide bonds overwhelmingly adopt the trans isomer (typically 99.9% under unstrained conditions), chiefly because the amide hydrogen (trans isomer) offers less steric repulsion to the preceding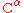 atom than does the following
atom than does the following 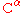 atom (cis isomer). By contrast, the cis and trans isomers of the X-Pro peptide bond (where X represents any amino acid) both experience steric clashes with the neighboring substitution and are nearly equal energetically. Hence, the fraction of X-Pro peptide bonds in the cis isomer under unstrained conditions ranges from 10-40%; the fraction depends slightly on the preceding amino acid, with aromatic residues favoring the cis isomer slightly.
atom (cis isomer). By contrast, the cis and trans isomers of the X-Pro peptide bond (where X represents any amino acid) both experience steric clashes with the neighboring substitution and are nearly equal energetically. Hence, the fraction of X-Pro peptide bonds in the cis isomer under unstrained conditions ranges from 10-40%; the fraction depends slightly on the preceding amino acid, with aromatic residues favoring the cis isomer slightly.
From a kinetic standpoint, cis-trans proline isomer
ization is a very slow process that can impede the progress of protein folding
by trapping one or more proline residues crucial for folding in the non-native isomer, especially when the native protein requires the cis isomer. This is because proline residues are exclusively synthesized in the ribosome
as the trans isomer form. All organisms possess prolyl isomerase
enzyme
s to catalyze this isomerization, and some bacteria
have specialized prolyl isomerases associated with the ribosome. However, not all prolines are essential for folding, and protein folding may proceed at a normal rate despite having non-native conformers of many X-Pro peptide bonds.
and proline catalysed aldol condensation
are prominent examples.
L-Proline is an osmoprotectant
and therefore is used in many pharmaceutical, biotechnological applications.
In brewing, proteins rich in proline combine with polyphenols to produce haze (turbidity).
, along with glycine
. Due to the ring formation connected to the Beta-carbon, the ψ and φ angles about the peptide bond have less allowable degrees of rotation. As a result it is often found in "turns" of proteins as its free entropy (ΔS) is not as comparatively large to other amino acids and thus in a folded form vs. unfolded form, the change in entropy is less. Furthermore, proline is rarely found in α and β structures as it would reduce the stability of such structures, because its side chain α-N can only form one hydrogen bond.
Additionally, proline is the only amino acid that does not form a blue/purple colour when developed by spraying with ninhydrin
for uses in chromatography
. Proline, instead, produces an orange/yellow colour.
synthesized proline by the reaction of sodium salt of diethyl malonate
with 1,3-Dibromopropane
in 1900. In 1901, Hermann Emil Fischer
isolated proline from casein and the decomposition products of γ-phthalimido-propylmalonic ester.
Amino acid
Amino acids are molecules containing an amine group, a carboxylic acid group and a side-chain that varies between different amino acids. The key elements of an amino acid are carbon, hydrogen, oxygen, and nitrogen...
, one of the twenty DNA
DNA
Deoxyribonucleic acid is a nucleic acid that contains the genetic instructions used in the development and functioning of all known living organisms . The DNA segments that carry this genetic information are called genes, but other DNA sequences have structural purposes, or are involved in...
-encoded amino acids. Its codons are CCU, CCC, CCA, and CCG. It is not an essential amino acid
Essential amino acid
An essential amino acid or indispensable amino acid is an amino acid that cannot be synthesized de novo by the organism , and therefore must be supplied in the diet.-Essentiality vs. conditional essentiality in humans:...
, which means that the human body can synthesize it. It is unique among the 20 protein-forming amino acids in that the α-amino group is secondary. The more common L form has S stereochemistry.
Biosynthesis
Proline is biosyntheticallyBiosynthesis
Biosynthesis is an enzyme-catalyzed process in cells of living organisms by which substrates are converted to more complex products. The biosynthesis process often consists of several enzymatic steps in which the product of one step is used as substrate in the following step...
derived from the amino acid L-glutamate and its immediate precursor is the imino acid
Imino acid
In chemistry, an imino acid is any molecule that contains both imino and carboxyl functional groups.Imino acids are related to amino acids, which contain both amino and carboxyl functional groups...
(S)-1-pyrroline-5-carboxylate
1-Pyrroline-5-carboxylic acid
1-Pyrroline-5-carboxylic acid, also known as 1-pyrroline-5-carboxylate, delta-1-pyrroline-5-carboxylic acid, and P5C, is an imino acid...
(P5C). Enzymes involved in a typical biosynthesis include:
- Glutamate 5-kinaseGlutamate 5-kinaseIn enzymology, a glutamate 5-kinase is an enzyme that catalyzes the chemical reactionThus, the two substrates of this enzyme are ATP and L-glutamate, whereas its two products are ADP and L-glutamate 5-phosphate....
, Glutamate 1-kinaseGlutamate 1-kinaseIn enzymology, a glutamate 1-kinase is an enzyme that catalyzes the chemical reactionThus, the two substrates of this enzyme are ATP and L-glutamate, whereas its two products are ADP and alpha-L-glutamyl phosphate....
(ATP-dependent) - Glutamate dehydrogenaseGlutamate dehydrogenaseGlutamate dehydrogenase is an enzyme, present in most microbes and the mitochondria of eukaryotes, as are some of the other enzymes required for urea synthesis, that converts glutamate to α-Ketoglutarate, and vice versa. In animals, the produced ammonia is, however, usually bled off to the urea...
(requires NADH or NADPH) - Pyrroline-5-carboxylate reductasePyrroline-5-carboxylate reductaseIn enzymology, a pyrroline-5-carboxylate reductase is an enzyme that catalyzes the chemical reactionThe 3 substrates of this enzyme are L-proline, NAD+, and NADP+, whereas its 4 products are 1-pyrroline-5-carboxylate, NADH, NADPH, and H+....
(requires NADH or NADPH)
Structural properties
The distinctive cyclic structure of proline's side chain locks its φ backbone dihedral angleDihedral angle
In geometry, a dihedral or torsion angle is the angle between two planes.The dihedral angle of two planes can be seen by looking at the planes "edge on", i.e., along their line of intersection...
at approximately −75°, giving proline an exceptional conformational rigidity compared to other amino acids. Hence, proline loses less conformational entropy
Introduction to entropy
Entropy is a measure of how evenly energy is distributed in a system. In a physical system, entropy provides a measure of the amount of energy that cannot be used to do work....
upon folding, which may account for its higher prevalence in the proteins of thermophilic
Thermophile
A thermophile is an organism — a type of extremophile — that thrives at relatively high temperatures, between 45 and 122 °C . Many thermophiles are archaea...
organisms. Proline acts as a structural disruptor in the middle of regular secondary structure
Secondary structure
In biochemistry and structural biology, secondary structure is the general three-dimensional form of local segments of biopolymers such as proteins and nucleic acids...
elements such as alpha helices
Alpha helix
A common motif in the secondary structure of proteins, the alpha helix is a right-handed coiled or spiral conformation, in which every backbone N-H group donates a hydrogen bond to the backbone C=O group of the amino acid four residues earlier...
and beta sheet
Beta sheet
The β sheet is the second form of regular secondary structure in proteins, only somewhat less common than the alpha helix. Beta sheets consist of beta strands connected laterally by at least two or three backbone hydrogen bonds, forming a generally twisted, pleated sheet...
s; however, proline is commonly found as the first residue of an alpha helix
Alpha helix
A common motif in the secondary structure of proteins, the alpha helix is a right-handed coiled or spiral conformation, in which every backbone N-H group donates a hydrogen bond to the backbone C=O group of the amino acid four residues earlier...
and also in the edge strands of beta sheet
Beta sheet
The β sheet is the second form of regular secondary structure in proteins, only somewhat less common than the alpha helix. Beta sheets consist of beta strands connected laterally by at least two or three backbone hydrogen bonds, forming a generally twisted, pleated sheet...
s. Proline is also commonly found in turns
Turn (biochemistry)
A turn is an element of secondary structure in proteins where the polypeptide chain reverses its overall direction.- Definition :According to the most common definition, a turn is a structural motif where the Cα atoms of two residues separated by few peptide bonds are in close approach A turn is...
, which may account for the curious fact that proline is usually solvent-exposed, despite having a completely aliphatic side chain. Because proline lacks a hydrogen on the amide group, it cannot act as a hydrogen bond
Hydrogen bond
A hydrogen bond is the attractive interaction of a hydrogen atom with an electronegative atom, such as nitrogen, oxygen or fluorine, that comes from another molecule or chemical group. The hydrogen must be covalently bonded to another electronegative atom to create the bond...
donor, only as a hydrogen bond
Hydrogen bond
A hydrogen bond is the attractive interaction of a hydrogen atom with an electronegative atom, such as nitrogen, oxygen or fluorine, that comes from another molecule or chemical group. The hydrogen must be covalently bonded to another electronegative atom to create the bond...
acceptor.
The distinct side chain/amine group interactions allow proline to aid in the formation of beta turns and also explains why it is not apparent in alpha helices...

Hydroxyproline
-4-Hydroxyproline, or L-hydroxyproline , is a common non-proteinogenic amino acid, abbreviated as HYP, e.g., in Protein Data Bank.-Structure and discovery:...
s in a row can create a polyproline helix
Polyproline helix
A Polyproline Helix is a type of protein secondary structure, which occurs in proteins comprising repeating proline residues. A left-handed polyproline II helix is formed when sequential residues all adopt backbone dihedral angles of roughly and have trans isomers of their peptide bonds...
, the predominant secondary structure
Secondary structure
In biochemistry and structural biology, secondary structure is the general three-dimensional form of local segments of biopolymers such as proteins and nucleic acids...
in collagen
Collagen
Collagen is a group of naturally occurring proteins found in animals, especially in the flesh and connective tissues of mammals. It is the main component of connective tissue, and is the most abundant protein in mammals, making up about 25% to 35% of the whole-body protein content...
. The hydroxylation
Hydroxylation
Hydroxylation is a chemical process that introduces a hydroxyl group into an organic compound. In biochemistry, hydroxylation reactions are often facilitated by enzymes called hydroxylases. Hydroxylation is the first step in the oxidative degradation of organic compounds in air...
of proline by prolyl hydroxylase
Prolyl hydroxylase
Prolyl hydroxylase is an enzyme involved in the production of collagen, acting to hydroxylate proline to hydroxyproline....
(or other additions of electron-withdrawing substituents such as fluorine
Fluorine
Fluorine is the chemical element with atomic number 9, represented by the symbol F. It is the lightest element of the halogen column of the periodic table and has a single stable isotope, fluorine-19. At standard pressure and temperature, fluorine is a pale yellow gas composed of diatomic...
) increases the conformational stability of collagen
Collagen
Collagen is a group of naturally occurring proteins found in animals, especially in the flesh and connective tissues of mammals. It is the main component of connective tissue, and is the most abundant protein in mammals, making up about 25% to 35% of the whole-body protein content...
significantly. Hence, the hydroxylation of proline is a critical biochemical process for maintaining the connective tissue
Connective tissue
"Connective tissue" is a fibrous tissue. It is one of the four traditional classes of tissues . Connective Tissue is found throughout the body.In fact the whole framework of the skeleton and the different specialized connective tissues from the crown of the head to the toes determine the form of...
of higher organisms. Severe diseases such as scurvy
Scurvy
Scurvy is a disease resulting from a deficiency of vitamin C, which is required for the synthesis of collagen in humans. The chemical name for vitamin C, ascorbic acid, is derived from the Latin name of scurvy, scorbutus, which also provides the adjective scorbutic...
can result from defects in this hydroxylation, e.g., mutations in the enzyme prolyl hydroxylase
Hydroxylation
Hydroxylation is a chemical process that introduces a hydroxyl group into an organic compound. In biochemistry, hydroxylation reactions are often facilitated by enzymes called hydroxylases. Hydroxylation is the first step in the oxidative degradation of organic compounds in air...
or lack of the necessary ascorbate (vitamin C)
Vitamin C
Vitamin C or L-ascorbic acid or L-ascorbate is an essential nutrient for humans and certain other animal species. In living organisms ascorbate acts as an antioxidant by protecting the body against oxidative stress...
cofactor.
Sequences of proline and 2-aminoisobutyric acid
2-Aminoisobutyric acid
2-Aminoisobutyric acid, or α-aminoisobutyric acid or α-methylalanine or 2-methylalanine, is an amino acid with the structural formula is H2N-C2-COOH. It is contained in some antibiotics of fungal origin, e.g. alamethicin and some lantibiotics. It is not one of the proteinogenic amino acids and...
(Aib) also form a helical turn structure.
In 2006, scientists at ASU
Arizona State University
Arizona State University is a public research university located in the Phoenix Metropolitan Area of the State of Arizona...
discovered that solutions of TiO2
Titanium dioxide
Titanium dioxide, also known as titanium oxide or titania, is the naturally occurring oxide of titanium, chemical formula . When used as a pigment, it is called titanium white, Pigment White 6, or CI 77891. Generally it comes in two different forms, rutile and anatase. It has a wide range of...
illuminated with ultraviolet
Ultraviolet
Ultraviolet light is electromagnetic radiation with a wavelength shorter than that of visible light, but longer than X-rays, in the range 10 nm to 400 nm, and energies from 3 eV to 124 eV...
radiation can serve as an extremely cost-effective and accurate protein cleavage catalyst. The TiO2 catalyst preferentially and rapidly cleaves protein at sites where proline is present, while taking much longer to degrade the protein from its endpoints.
Peptide bond formation with incoming Pro-tRNAPro is considerably slower than with any other tRNAs, which is a general feature of N-alkylamino acids.
Cis-trans isomerization
Peptide bondPeptide bond
This article is about the peptide link found within biological molecules, such as proteins. A similar article for synthetic molecules is being created...
s to proline, and to other N-substituted amino acids (such as sarcosine
Sarcosine
Sarcosine, also known as N-methylglycine, is an intermediate and byproduct in glycine synthesis and degradation. Sarcosine is metabolized to glycine by the enzyme sarcosine dehydrogenase, while glycine-N-methyl transferase generates sarcosine from glycine. Sarcosine is a natural amino acid found in...
), are able to populate both the cis and trans isomers. Most peptide bonds overwhelmingly adopt the trans isomer (typically 99.9% under unstrained conditions), chiefly because the amide hydrogen (trans isomer) offers less steric repulsion to the preceding
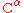 atom than does the following
atom than does the following 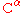 atom (cis isomer). By contrast, the cis and trans isomers of the X-Pro peptide bond (where X represents any amino acid) both experience steric clashes with the neighboring substitution and are nearly equal energetically. Hence, the fraction of X-Pro peptide bonds in the cis isomer under unstrained conditions ranges from 10-40%; the fraction depends slightly on the preceding amino acid, with aromatic residues favoring the cis isomer slightly.
atom (cis isomer). By contrast, the cis and trans isomers of the X-Pro peptide bond (where X represents any amino acid) both experience steric clashes with the neighboring substitution and are nearly equal energetically. Hence, the fraction of X-Pro peptide bonds in the cis isomer under unstrained conditions ranges from 10-40%; the fraction depends slightly on the preceding amino acid, with aromatic residues favoring the cis isomer slightly.From a kinetic standpoint, cis-trans proline isomer
Isomer
In chemistry, isomers are compounds with the same molecular formula but different structural formulas. Isomers do not necessarily share similar properties, unless they also have the same functional groups. There are many different classes of isomers, like stereoisomers, enantiomers, geometrical...
ization is a very slow process that can impede the progress of protein folding
Protein folding
Protein folding is the process by which a protein structure assumes its functional shape or conformation. It is the physical process by which a polypeptide folds into its characteristic and functional three-dimensional structure from random coil....
by trapping one or more proline residues crucial for folding in the non-native isomer, especially when the native protein requires the cis isomer. This is because proline residues are exclusively synthesized in the ribosome
Ribosome
A ribosome is a component of cells that assembles the twenty specific amino acid molecules to form the particular protein molecule determined by the nucleotide sequence of an RNA molecule....
as the trans isomer form. All organisms possess prolyl isomerase
Prolyl isomerase
Prolyl isomerase is an enzyme found in both prokaryotes and eukaryotes that interconverts the cis and trans isomers of peptide bonds with the amino acid proline. Proline has an unusually conformationally restrained peptide bond due to its cyclic structure with its side chain bonded to its...
enzyme
Enzyme
Enzymes are proteins that catalyze chemical reactions. In enzymatic reactions, the molecules at the beginning of the process, called substrates, are converted into different molecules, called products. Almost all chemical reactions in a biological cell need enzymes in order to occur at rates...
s to catalyze this isomerization, and some bacteria
Bacteria
Bacteria are a large domain of prokaryotic microorganisms. Typically a few micrometres in length, bacteria have a wide range of shapes, ranging from spheres to rods and spirals...
have specialized prolyl isomerases associated with the ribosome. However, not all prolines are essential for folding, and protein folding may proceed at a normal rate despite having non-native conformers of many X-Pro peptide bonds.
Uses
Proline and its derivatives are often used as asymmetric catalysts in organic reactions. The CBS reductionCBS reduction
The Corey-Itsuno Reduction, also known as the Corey-Bakshi-Shibata reduction, is a chemical reaction in which an achiral ketone is enantioselectively reduced to produce the corresponding chiral, non-racemic alcohol...
and proline catalysed aldol condensation
Aldol condensation
An aldol condensation is an organic reaction in which an enol or an enolate ion reacts with a carbonyl compound to form a β-hydroxyaldehyde or β-hydroxyketone, followed by a dehydration to give a conjugated enone....
are prominent examples.
L-Proline is an osmoprotectant
Osmoprotectant
Osmoprotectants or compatible solutes are small molecules that act as osmolytes and help organisms survive extreme osmotic stress. Examples include betaines, amino acids, and the sugar trehalose. These molecules accumulate in cells and balance the osmotic difference between the cell's surroundings...
and therefore is used in many pharmaceutical, biotechnological applications.
In brewing, proteins rich in proline combine with polyphenols to produce haze (turbidity).
Specialities
Proline is one of the two amino acids that do not follow along with the typical Ramachandran plotRamachandran plot
-Introduction and early history:A Ramachandran plot , originally developed in 1963 by G. N. Ramachandran C. Ramakrishnan and V...
, along with glycine
Glycine
Glycine is an organic compound with the formula NH2CH2COOH. Having a hydrogen substituent as its 'side chain', glycine is the smallest of the 20 amino acids commonly found in proteins. Its codons are GGU, GGC, GGA, GGG cf. the genetic code.Glycine is a colourless, sweet-tasting crystalline solid...
. Due to the ring formation connected to the Beta-carbon, the ψ and φ angles about the peptide bond have less allowable degrees of rotation. As a result it is often found in "turns" of proteins as its free entropy (ΔS) is not as comparatively large to other amino acids and thus in a folded form vs. unfolded form, the change in entropy is less. Furthermore, proline is rarely found in α and β structures as it would reduce the stability of such structures, because its side chain α-N can only form one hydrogen bond.
Additionally, proline is the only amino acid that does not form a blue/purple colour when developed by spraying with ninhydrin
Ninhydrin
Ninhydrin is a chemical used to detect ammonia or primary and secondary amines. When reacting with these free amines, a deep blue or purple color known as Ruhemann's purple is produced...
for uses in chromatography
Chromatography
Chromatography is the collective term for a set of laboratory techniques for the separation of mixtures....
. Proline, instead, produces an orange/yellow colour.
History
Richard WillstätterRichard Willstätter
Richard Martin Willstätter was a German organic chemist whose study of the structure of plant pigments, chlorophyll included, won him the 1915 Nobel Prize for Chemistry. Willstätter invented paper chromatography independently of Mikhail Tsvet.-Biography:Willstätter was born in to a Jewish family...
synthesized proline by the reaction of sodium salt of diethyl malonate
Diethyl malonate
Diethyl malonate, also known as DEM, is the diethyl ester of malonic acid. It occurs naturally in grapes and strawberries as a colourless liquid with an apple-like odour, and is used in perfumes...
with 1,3-Dibromopropane
1,3-Dibromopropane
1,3-Dibromopropane is a halogenated hydrocarbon. When at room temperature, it is a colorless to light-brown liquid. Synthetically, it is very useful to form C3-bridged compounds such as through C-N coupling reactions....
in 1900. In 1901, Hermann Emil Fischer
Hermann Emil Fischer
Hermann Emil Fischer, Emil Fischer was a German chemist and 1902 recipient of the Nobel Prize in Chemistry. He discovered the Fischer esterification. He developed the Fischer projection, a symbolic way of drawing asymmetric carbon atoms.-Early years:Fischer was born in Euskirchen, near Cologne,...
isolated proline from casein and the decomposition products of γ-phthalimido-propylmalonic ester.
See also
- ProlinolProlinolProlinol is a chiral amino-alcohol that is used as a chiral building block in organic synthesis. It exists as two enantiomers: the D and L forms.-Preparation:Prolinol is obtained by reduction of the amino acid proline using lithium aluminium hydride...
- CollagenCollagenCollagen is a group of naturally occurring proteins found in animals, especially in the flesh and connective tissues of mammals. It is the main component of connective tissue, and is the most abundant protein in mammals, making up about 25% to 35% of the whole-body protein content...
- Polyproline helixPolyproline helixA Polyproline Helix is a type of protein secondary structure, which occurs in proteins comprising repeating proline residues. A left-handed polyproline II helix is formed when sequential residues all adopt backbone dihedral angles of roughly and have trans isomers of their peptide bonds...
- Peptide bondPeptide bondThis article is about the peptide link found within biological molecules, such as proteins. A similar article for synthetic molecules is being created...
(For more discussion of cis-trans isomerization) - HyperprolinemiaHyperprolinemiaHyperprolinemia, also referred to as prolinemia or prolinuria, is a condition which occurs when the amino acid proline is not broken down properly by the enzymes proline oxidase or pyrroline-5-carboxylate dehydrogense, causing a build up of proline in the body.-Hyperprolinemia type I:It is...
Synthesis
The depicted scheme is for racemic proline and was taken from Vogel Practical Organic Chemistry 5th edition.Further reading
.- For a thorough scientific overview of disorders of proline and hydroxyproline metabolism, one can consult chapter 81 of OMMBID Charles ScriverCharles ScriverCharles Robert Scriver, is an eminent Canadian pediatrician and biochemical geneticist. Scriver made many important contributions to our knowledge of inborn errors of metabolism...
, Beaudet, A.L., Valle, D., Sly, W.S., Vogelstein, B., Childs, B., Kinzler, K.W. (Accessed 2007). The Online Metabolic and Molecular Bases of Inherited Disease. New York: McGraw-Hill. - Summaries of 255 chapters, full text through many universities. There is also the OMMBID blog. - For more online resources and references, see inborn errors of metabolism.

