Crystallographic defect
Encyclopedia
Crystalline solids exhibit a periodic crystal structure
. The positions of atoms or molecules occur on repeating fixed distances, determined by the unit cell parameters. However, the arrangement of atom or molecules in most crystalline materials is not perfect. The regular patterns are interrupted by crystallographic defects.
loops. For historical reasons, many point defects, especially in ionic crystals, are called centers: for example a vacancy in many ionic solids is called a luminescence center, a color center, or F-center
. These dislocations permit ionic transport through crystals leading to electrochemical reactions. These are frequently specified using Kröger–Vink Notation.
There are two basic types of dislocations, the edge dislocation and the screw dislocation. "Mixed" dislocations, combining aspects of both types, are also common.
Edge dislocations are caused by the termination of a plane of atoms in the middle of a crystal. In such a case, the adjacent planes are not straight, but instead bend around the edge of the terminating plane so that the crystal structure is perfectly ordered on either side. The analogy with a stack of paper is apt: if a half a piece of paper is inserted in a stack of paper, the defect in the stack is only noticeable at the edge of the half sheet.
The screw dislocation is more difficult to visualise, but basically comprises a structure in which a helical path is traced around the linear defect (dislocation line) by the atomic planes of atoms in the crystal lattice.
The presence of dislocation results in lattice strain (distortion). The direction and magnitude of such distortion is expressed in terms of a Burgers vector
(b). For an edge type, b is perpendicular to the dislocation line, whereas in the cases of the screw type it is parallel. In metallic materials, b is aligned with close-packed crytallographic directions and its magnitude is equivalent to one interatomic spacing.
Dislocations can move if the atoms from one of the surrounding planes break their bonds and rebond with the atoms at the terminating edge.
It is the presence of dislocations and their ability to readily move (and interact) under the influence of stresses induced by external loads that leads to the characteristic malleability of metallic materials.
Dislocations can be observed using transmission electron microscopy
, field ion microscopy and atom probe
techniques.
Deep level transient spectroscopy has been used for studying the electrical activity of dislocations in semiconductors, mainly silicon
.
s in liquid crystals and for excitations in superfluid 3He, is the topological homotopy
theory.
and/or in containers with non-commeasurable sizes
using the Lubachevsky-Stillinger algorithm
can be an effective techniques for demonstrating
some types of crystallographic defects.
Crystal structure
In mineralogy and crystallography, crystal structure is a unique arrangement of atoms or molecules in a crystalline liquid or solid. A crystal structure is composed of a pattern, a set of atoms arranged in a particular way, and a lattice exhibiting long-range order and symmetry...
. The positions of atoms or molecules occur on repeating fixed distances, determined by the unit cell parameters. However, the arrangement of atom or molecules in most crystalline materials is not perfect. The regular patterns are interrupted by crystallographic defects.
Point defects
Point defects are defects that occur only at or around a single lattice point. They are not extended in space in any dimension. Strict limits for how small a point defect is, are generally not defined explicitly, but typically these defects involve at most a few extra or missing atoms. Larger defects in an ordered structure are usually considered dislocationDislocation
In materials science, a dislocation is a crystallographic defect, or irregularity, within a crystal structure. The presence of dislocations strongly influences many of the properties of materials...
loops. For historical reasons, many point defects, especially in ionic crystals, are called centers: for example a vacancy in many ionic solids is called a luminescence center, a color center, or F-center
F-Center
An F-Center or Farbe center is a type of crystallographic defect in which an anionic vacancy in a crystal is filled by one or more electrons, depending on the charge of the missing ion in the crystal. Electrons in such a vacancy tend to absorb light in the visible spectrum such that a material...
. These dislocations permit ionic transport through crystals leading to electrochemical reactions. These are frequently specified using Kröger–Vink Notation.
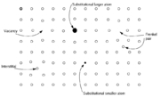
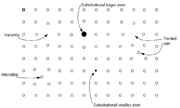
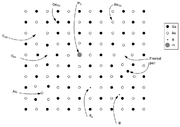
- Vacancy defects are lattice sites which would be occupied in a perfect crystal, but are vacant. If a neighboring atom moves to occupy the vacant site, the vacancy moves in the opposite direction to the site which used to be occupied by the moving atom. The stability of the surrounding crystal structure guarantees that the neighboring atoms will not simply collapse around the vacancy. In some materials, neighboring atoms actually move away from a vacancy, because they experience attraction from atoms in the surroundings. A vacancy (or pair of vacancies in an ionic solid) is sometimes called a Schottky defectSchottky defectA Schottky defect is a type of point defect in a crystal lattice named after Walter H. Schottky.The defect forms when oppositely charged ions leave their lattice sites, creating vacancies. These vacancies are formed in stoichiometric units, to maintain an overall neutral charge in the ionic solid....
.
- Interstitial defectInterstitial defectInterstitials are a variety of crystallographic defects, i.e. atoms which occupy a site in the crystal structure at which there is usually not an atom, or two or more atoms sharing one or more lattice sites such that the number of atoms is larger than the number of lattice sites.They are generally...
s are atoms that occupy a site in the crystal structure at which there is usually not an atom. They are generally high energy configurations. Small atoms in some crystals can occupy interstices without high energy, such as hydrogenHydrogenHydrogen is the chemical element with atomic number 1. It is represented by the symbol H. With an average atomic weight of , hydrogen is the lightest and most abundant chemical element, constituting roughly 75% of the Universe's chemical elemental mass. Stars in the main sequence are mainly...
in palladiumPalladiumPalladium is a chemical element with the chemical symbol Pd and an atomic number of 46. It is a rare and lustrous silvery-white metal discovered in 1803 by William Hyde Wollaston. He named it after the asteroid Pallas, which was itself named after the epithet of the Greek goddess Athena, acquired...
.

- A nearby pair of a vacancy and an interstitial is often called a Frenkel defectFrenkel defectThe Frenkel Defect is shown by ionic solids. The smaller ion is displaced from its lattice position to an interstitial site. It creates a vacancy defect at its original site and an interstitial defect at its new location.-Definition:...
or Frenkel pair. This is caused when an ion moves into an interstitial site and creates a vacancy.
- Impurities occur because materials are never 100% pure. In the case of an impurity, the atom is often incorporated at a regular atomic site in the crystal structure. This is neither a vacant site nor is the atom on an interstitial site and it is called a substitutional defect. The atom is not supposed to be anywhere in the crystal, and is thus an impurity. There are two different types of substitutional defects. Isovalent substitution and aliovalent substitution. Isovalent substitution is where the ion that is substituting the original ion is of the same oxidation state as the ion it is replacing. Aliovalent substitution is where the ion that is substituting the original ion is of a different oxidation state as the ion it is replacing. Aliovalent substitutions change the overall charge within the ionic compound, but the ionic compound must be neutral. Therefore a charge compensation mechanism is required. Hence either one of the metals is partially or fully oxidised or reduced, or ion vacancies are created.
- Antisite defects occur in an ordered alloy or compound when atoms of different type exchange positions. For example, some alloys have a regular structure in which every other atom is a different species; for illustration assume that type A atoms sit on the corners of a cubic lattice, and type B atoms sit in the center of the cubes. If one cube has an A atom at its center, the atom is on a site usually occupied by a B atom, and is thus an antisite defect. This is neither a vacancy nor an interstitial, nor an impurity.
- Topological defects are regions in a crystal where the normal chemical bonding environment is topologically different from the surroundings. For instance, in a perfect sheet of graphite (grapheneGrapheneGraphene is an allotrope of carbon, whose structure is one-atom-thick planar sheets of sp2-bonded carbon atoms that are densely packed in a honeycomb crystal lattice. The term graphene was coined as a combination of graphite and the suffix -ene by Hanns-Peter Boehm, who described single-layer...
) all atoms are in rings containing six atoms. If the sheet contains regions where the number of atoms in a ring is different from six, while the total number of atoms remains the same, a topological defect has formed. An example is the Stone Wales defectStone Wales defectA Stone–Wales defect is a crystallographic defect that occurs on carbon nanotubes and graphene and is thought to have important implications for nanotube's mechanical properties. The defect is named after Anthony Stone and David Wales of Cambridge University, who described it in a 1986 paper on the...
in nanotubes, which consists of two adjacent 5-membered and two 7-membered atom rings.

- Also amorphous solids may contain defects. These are naturally somewhat hard to define, but sometimes their nature can be quite easily understood. For instance, in ideally bonded amorphous silica all Si atoms have 4 bonds to O atoms and all O atoms have 2 bonds to Si atom. Thus e.g. an O atom with only one Si bond (a dangling bondDangling bondIn chemistry, a dangling bond is an unsatisfied valence on an immobilised atom.In order to gain enough electrons to fill their valence shells , many atoms will form covalent bonds with other atoms. In the simplest case, that of a single bond, two atoms each contribute one unpaired electron, and the...
) can be considered a defect in silica.
- Complexes can form between different kinds of point defects. For example, if a vacancy encounters an impurity, the two may bind together if the impurity is too large for the lattice. Interstitials can form 'split interstitial' or 'dumbbell' structures where two atoms effectively share an atomic site, resulting in neither atom actually occupying the site.
Line defects
Line defects can be described by gauge theories.- DislocationDislocationIn materials science, a dislocation is a crystallographic defect, or irregularity, within a crystal structure. The presence of dislocations strongly influences many of the properties of materials...
s are linear defects around which some of the atoms of the crystal lattice are misaligned.
There are two basic types of dislocations, the edge dislocation and the screw dislocation. "Mixed" dislocations, combining aspects of both types, are also common.
Edge dislocations are caused by the termination of a plane of atoms in the middle of a crystal. In such a case, the adjacent planes are not straight, but instead bend around the edge of the terminating plane so that the crystal structure is perfectly ordered on either side. The analogy with a stack of paper is apt: if a half a piece of paper is inserted in a stack of paper, the defect in the stack is only noticeable at the edge of the half sheet.
The screw dislocation is more difficult to visualise, but basically comprises a structure in which a helical path is traced around the linear defect (dislocation line) by the atomic planes of atoms in the crystal lattice.
The presence of dislocation results in lattice strain (distortion). The direction and magnitude of such distortion is expressed in terms of a Burgers vector
Burgers vector
The Burgers vector, named after Dutch physicist Jan Burgers, is a vector, often denoted b, that represents the magnitude and direction of the lattice distortion of dislocation in a crystal lattice....
(b). For an edge type, b is perpendicular to the dislocation line, whereas in the cases of the screw type it is parallel. In metallic materials, b is aligned with close-packed crytallographic directions and its magnitude is equivalent to one interatomic spacing.
Dislocations can move if the atoms from one of the surrounding planes break their bonds and rebond with the atoms at the terminating edge.
It is the presence of dislocations and their ability to readily move (and interact) under the influence of stresses induced by external loads that leads to the characteristic malleability of metallic materials.
Dislocations can be observed using transmission electron microscopy
Transmission electron microscopy
Transmission electron microscopy is a microscopy technique whereby a beam of electrons is transmitted through an ultra thin specimen, interacting with the specimen as it passes through...
, field ion microscopy and atom probe
Atom probe
The atom probe is a microscope used in material science that was invented in 1967 by Erwin Wilhelm Müller, J. A. Panitz, and S. Brooks McLane. The atom probe is closely related to the method of Field Ion Microscopy, which is the first microscopic method to achieve atomic resolution, occurring in...
techniques.
Deep level transient spectroscopy has been used for studying the electrical activity of dislocations in semiconductors, mainly silicon
Silicon
Silicon is a chemical element with the symbol Si and atomic number 14. A tetravalent metalloid, it is less reactive than its chemical analog carbon, the nonmetal directly above it in the periodic table, but more reactive than germanium, the metalloid directly below it in the table...
.
- DisclinationDisclinationA disclination is a line defect in which rotational symmetry is violated. In analogy with dislocations in crystals, the term, disinclination, for liquid crystals first used by F. C. Frank and since then has been modified to its current usage, disclination.It is a defect in the orientation of...
s are line defects corresponding to "adding" or "subtracting" an angle around a line. Basically, this means that if you track the crystal orientation around the line defect, you get a rotation. Usually they play a role only in liquid crystals.
Planar defects
- Grain boundaries occur where the crystallographic direction of the lattice abruptly changes. This usually occurs when two crystals begin growing separately and then meet.
- Antiphase boundaries occur in ordered alloys: in this case, the crystallographic direction remains the same, but each side of the boundary has an opposite phase: For example, if the ordering is usually ABABABAB, an antiphase boundary takes the form of ABABBABA.
- Stacking faults occur in a number of crystal structures, but the common example is in close-packedClose-packingIn geometry, close-packing of equal spheres is a dense arrangement of congruent spheres in an infinite, regular arrangement . Carl Friedrich Gauss proved that the highest average density – that is, the greatest fraction of space occupied by spheres – that can be achieved by a regular lattice...
structures. Face-centered cubic (fcc) structures differ from hexagonal close packed (hcp) structures only in stacking order: both structures have close packed atomic planes with sixfold symmetry—the atoms form equilateral triangles. When stacking one of these layers on top of another, the atoms are not directly on top of one another—the first two layers are identical for hcp and fcc, and labelled AB. If the third layer is placed so that its atoms are directly above those of the first layer, the stacking will be ABA—this is the hcp structure, and it continues ABABABAB. However, there is another possible location for the third layer, such that its atoms are not above the first layer. Instead, it is the atoms in the fourth layer that are directly above the first layer. This produces the stacking ABCABCABC, and is actually a cubic arrangement of the atoms. A stacking fault is a one or two layer interruption in the stacking sequence, for example, if the sequence ABCABABCAB were found in an fcc structure.
Bulk defects
- Voids are small regions where there are no atoms, and can be thought of as clusters of vacancies.
- Impurities can cluster together to form small regions of a different phase. These are often called precipitates.
Mathematical classification methods
A successful mathematical classification method for physical lattice defects, which works not only with the theory of dislocations and other defects in crystals but also, e.g., for disclinationDisclination
A disclination is a line defect in which rotational symmetry is violated. In analogy with dislocations in crystals, the term, disinclination, for liquid crystals first used by F. C. Frank and since then has been modified to its current usage, disclination.It is a defect in the orientation of...
s in liquid crystals and for excitations in superfluid 3He, is the topological homotopy
Homotopy
In topology, two continuous functions from one topological space to another are called homotopic if one can be "continuously deformed" into the other, such a deformation being called a homotopy between the two functions...
theory.
Computer simulation methods
Simulating jamming of hard spheres of different sizesand/or in containers with non-commeasurable sizes
using the Lubachevsky-Stillinger algorithm
Lubachevsky-Stillinger algorithm
Lubachevsky-Stillinger algorithm isa numerical procedure that simulates or imitatesa physical process of compressing an assemblyof hard particles...
can be an effective techniques for demonstrating
some types of crystallographic defects.
See also
- Bjerrum defectBjerrum defectA Bjerrum defect is a crystallographic defect which is specific to ice, and which is partly responsible for the electrical properties of ice. A hydrogen bond normally has one proton, but a hydrogen bond with a Bjerrum defect will have either two protons or no proton...
- Crystallographic defects in diamondCrystallographic defects in diamondImperfections in the crystal lattice of diamond are common. Such crystallographic defects in diamond may be the result of lattice irregularities or extrinsic substitutional or interstitial impurities, introduced during or after the diamond growth...
- Kröger-Vink NotationKröger-Vink NotationKröger–Vink notation is set of conventions used to describe electric charge and lattice position for point defect species in crystals. It is primarily used for ionic crystals and is particularly useful for describing various defect reactions. It was proposed by F. A. Kröger and H. J. Vink.-General...
- F-CenterF-CenterAn F-Center or Farbe center is a type of crystallographic defect in which an anionic vacancy in a crystal is filled by one or more electrons, depending on the charge of the missing ion in the crystal. Electrons in such a vacancy tend to absorb light in the visible spectrum such that a material...
Further reading
- Hagen KleinertHagen KleinertHagen Kleinert is Professor of Theoretical Physics at the Free University of Berlin, Germany , at theWest University of Timişoara, at thein Bishkek. He is also of the...
, Gauge Fields in Condensed Matter, Vol. II, "Stresses and defects", pp. 743–1456, World Scientific (Singapore, 1989); Paperback ISBN 9971-5-0210-0 - Hermann Schmalzried: Solid State Reactions. Verlag Chemie, Weinheim 1981, ISBN 3-527-25872-8.

