
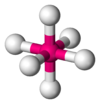
Octahedral molecular geometry
Encyclopedia
Chemistry
Chemistry is the science of matter, especially its chemical reactions, but also its composition, structure and properties. Chemistry is concerned with atoms and their interactions with other atoms, and particularly with the properties of chemical bonds....
, octahedral molecular geometry describes the shape of compounds where in six atoms or groups of atoms or ligand
Ligand
In coordination chemistry, a ligand is an ion or molecule that binds to a central metal atom to form a coordination complex. The bonding between metal and ligand generally involves formal donation of one or more of the ligand's electron pairs. The nature of metal-ligand bonding can range from...
s are symmetrically arranged around a central atom, defining the vertices of an octahedron. The octahedron
Octahedron
In geometry, an octahedron is a polyhedron with eight faces. A regular octahedron is a Platonic solid composed of eight equilateral triangles, four of which meet at each vertex....
has eight faces, hence the prefix octa. The octahedron is one of the Platonic solid
Platonic solid
In geometry, a Platonic solid is a convex polyhedron that is regular, in the sense of a regular polygon. Specifically, the faces of a Platonic solid are congruent regular polygons, with the same number of faces meeting at each vertex; thus, all its edges are congruent, as are its vertices and...
s, although octahedral molecules typically have an atom in their centre and no bonds between the ligand atoms. A perfect octahedron
Octahedron
In geometry, an octahedron is a polyhedron with eight faces. A regular octahedron is a Platonic solid composed of eight equilateral triangles, four of which meet at each vertex....
belongs to the point group
Point group
In geometry, a point group is a group of geometric symmetries that keep at least one point fixed. Point groups can exist in a Euclidean space with any dimension, and every point group in dimension d is a subgroup of the orthogonal group O...
Oh. Examples of octahedral compounds are sulfur hexafluoride
Sulfur hexafluoride
Sulfur hexafluoride is an inorganic, colorless, odorless, and non-flammable greenhouse gas. has an octahedral geometry, consisting of six fluorine atoms attached to a central sulfur atom. It is a hypervalent molecule. Typical for a nonpolar gas, it is poorly soluble in water but soluble in...
SF6 and molybdenum hexacarbonyl
Molybdenum hexacarbonyl
Molybdenum hexacarbonyl is the chemical compound with the formula Mo6. This colorless solid, like its chromium and tungsten analogues, is noteworthy as a volatile, air-stable derivative of a metal in its zero oxidation state.-Structure and properties:Mo6 adopts an octahedral geometry consisting...
Mo(CO)6. The term "octahedral" is used somewhat loosely by chemists: [Co(NH3)6]3+
Cobalt(III) hexammine chloride
Hexamminecobalt chloride is the chemical compound with the formula [Co6]Cl3. This coordination compound is considered an archetypal "Werner complex", named after the pioneer of coordination chemistry, Alfred Werner. This salt consists of [Co6]3+ trications with three Cl− anions...
, which is not octahedral in the mathematical sense due to the orientation of the N-H bonds, is referred to as octahedral.
The concept of octahedral coordination geometry was developed by Alfred Werner
Alfred Werner
Alfred Werner was a Swiss chemist who was a student at ETH Zurich and a professor at the University of Zurich. He won the Nobel Prize in Chemistry in 1913 for proposing the octahedral configuration of transition metal complexes. Werner developed the basis for modern coordination chemistry...
to explain the stoichiometries and isomerism in coordination compounds. His insight allowed chemists to rationalize the number of isomers of coordination compounds. Octahedral transition-metal complexes containing amines and simple anions are often referred to Werner-type complexes.
Isomerism in octahedral complexes
When two or more types of ligands are coordinated to an octahedral metal centre, the complex can exist as isomers. The naming system for these isomers depends upon the number and arrangement of different ligands.cis and trans
For MLa4Lb2, two isomers exist. These isomers of MLa4Lb2 are cis, if the Lb ligands are mutually adjacent, and trans, if the Lb groups are situated 180° to each other. It was the analysis of such complexes that led Alfred WernerAlfred Werner
Alfred Werner was a Swiss chemist who was a student at ETH Zurich and a professor at the University of Zurich. He won the Nobel Prize in Chemistry in 1913 for proposing the octahedral configuration of transition metal complexes. Werner developed the basis for modern coordination chemistry...
to the 1913 Nobel Prize winning postulation of octahedral complexes.
For MLa2Lb2Lc2, cis and trans isomers are also possible. All three types of ligands La, Lb and Lc may be trans, or one type may be trans while the other two are cis. This latter case gives two unique isomers (for a total of three).
Facial and meridional isomers
For MLa3Lb3, two isomers are possible - a facial isomer (fac) where the three identical ligands are mutually cis, and a meridional isomer (mer) where the three ligands are coplanar.Chirality
More complicated complexes, with several different kinds of ligands or with bidentate ligands can also be chiralChirality (chemistry)
A chiral molecule is a type of molecule that lacks an internal plane of symmetry and thus has a non-superimposable mirror image. The feature that is most often the cause of chirality in molecules is the presence of an asymmetric carbon atom....
.
Other
The number of possible isomers can reach 30 for an octahedral complex with six different ligands (in contrast, only two stereoisomers are possible for a tetrahedral complex with four different ligands). The following table lists all possible combinations for monodentate ligands:| Formula | Number of isomers | Number of enantiomeric pairs |
|---|---|---|
| ML6 | 1 | 0 |
| MLa5Lb | 1 | 0 |
| MLa4Lb2 | 2 | 0 |
| MLa4LbLc | 2 | 0 |
| MLa3Lb3 | 2 | 0 |
| MLa3Lb2Lc | 3 | 0 |
| MLa3LbLcLd | 5 | 1 |
| MLa2Lb2Lc2 | 6 | 1 |
| MLa2Lb2LcLd | 8 | 2 |
| MLa2LbLcLdLe | 15 | 6 |
| MLaLbLcLdLeLf | 30 | 15 |
Thus, all 15 diastereomers of MLaLbLcLdLeLf are chiral, whereas for MLa2LbLcLdLe, six diastereomers are chiral and three are not: the ones where La are trans. One can see that octahedral coordination allows much greater complexity
Complexity
In general usage, complexity tends to be used to characterize something with many parts in intricate arrangement. The study of these complex linkages is the main goal of complex systems theory. In science there are at this time a number of approaches to characterizing complexity, many of which are...
than the tetrahedron that dominates organic chemistry
Organic chemistry
Organic chemistry is a subdiscipline within chemistry involving the scientific study of the structure, properties, composition, reactions, and preparation of carbon-based compounds, hydrocarbons, and their derivatives...
. The tetrahedron MLaLbLcLd exists as a single enatiomeric pair. To generate two diastereomers in an organic compound, at least two carbon centers are required.
Trigonal prismatic geometry
For compounds with the formula MX6, the chief alternative to octahedral geometry is a trigonal prismatic geometry, which has symmetrySymmetry group
The symmetry group of an object is the group of all isometries under which it is invariant with composition as the operation...
D3h. In this geometry, the six ligands are also equivalent. There are also distorted trigonal prisms, with C3v symmetry; a prominent example is W(CH3)6
Hexamethyl tungsten
Hexamethyltungsten is the chemical compound W6. Classified as an organometallic compound, hexamethyltungsten is an air-sensitive, red, crystalline solid at room temperature; however, it is extremely volatile and sublimes at −30 °C...
. The interconversion of Δ- and Λ-complexes, which is usually slow, is proposed to proceed via a trigonal prismatic intermediate, a process called the "Bailar twist
Bailar twist
The Bailar twist is a mechanism proposed for the racemization of octahedral complexes containing three bidentate chelate rings. Such complexes typically adopt an octahedral molecular geometry and are thus chiral. One pathway by which these compounds can racemize is via the formation of a trigonal...
." An alternative pathway for the racemization of these same complexes is the Ray-Dutt twist
Ray-Dutt twist
The Ray-Dutt twist is a mechanism proposed for the racemization of octahedral complexes containing three bidentate chelate rings. Such complexes typically adopt an octahedral molecular geometry in their ground states and are thus chiral. The pathway entails formation of an intermediate of C2v...
.
Splitting of d-orbitals in octahedral complexes
For a "free ion", e.g. gaseous Ni2+ or Mo0, the d-orbitals are equi-energetic, that is they are "degenerate." In an octahedral complex, this degeneracy is lifted. The dz2 and dx2-y2, the so-called eg set, which are aimed directly at the ligands are destabilized. On the other hand, the dxz, dxy, and dyz orbitals, the so-called t2g set, are not. The labels t2g and eg refer to irreducible representations, which describe the symmetry properties of these orbitals. The energy gap separating these two sets is the basis of Crystal Field TheoryCrystal field theory
Crystal field theory is a model that describes the electronic structure of transition metal compounds, all of which can be considered coordination complexes. CFT successfully accounts for some magnetic properties, colours, hydration enthalpies, and spinel structures of transition metal complexes,...
and the more comprehensive Ligand Field Theory
Ligand field theory
Ligand field theory describes the bonding, orbital arrangement, and other characteristics of coordination complexes. It represents an application of molecular orbital theory to transition metal complexes. A transition metal ion has nine valence atomic orbitals, five d, one s, and three p orbitals...
. The loss of degeneracy upon the formation of an octahedral complex from a free ion is called crystal field splitting or ligand field splitting. The energy gap is labeled Δo, which varies according to the nature of the ligands. If the symmetry of the complex is lower than octahedral, the eg and t2g levels can split further. For example, the t2g and eg sets split further in trans-MLa4Lb2.
Ligand strength has the following order for these electron donors:
weak: iodine < bromine < fluorine < acetate < oxalate < water < pyridine < cyanide :strong
So called "weak field ligands" give rise to small Δo and absorb light at longer wavelength
Wavelength
In physics, the wavelength of a sinusoidal wave is the spatial period of the wave—the distance over which the wave's shape repeats.It is usually determined by considering the distance between consecutive corresponding points of the same phase, such as crests, troughs, or zero crossings, and is a...
s.
Reactions
Given that a virtually uncountable variety of octahedral complexes exist, it is not surprising that a wide variety of reactions have been described. These reactions can be classified as follows:- Ligand substitution reactions (via a variety of mechanisms)
- Ligand addition reactions, including among many, protonation
- Redox reactions (where electrons are gained or lost)
- Rearrangements where the relative stereochemistry of the ligand change within the coordination sphereCoordination sphereIn coordination chemistry, the coordination sphere refers to a central atom or ion and an array of molecules or anions, the ligands, around.Molecules that are attached noncovalently to the ligands are called the second coordination sphere....
.
Many reactions of octahedral transition metal complexes occur in water. When an anionic ligand replaces a coordinated water molecule the reaction is called an anation. The reverse reaction, water replacing an anionic ligand, is called an "aquation reaction." For example, the [Co(NH3)5Cl]2+ slowly aquates to give [Co(NH3)5(H2O)]3+ in water, especially in the presence of acid or base. Addition of concentrated HCl converts the aquo complex back to the chloride, via an anation process.
See also
- Octahedral clusterOctahedral clusterOctahedral clusters are inorganic or organometallic cluster compounds composed of six metals in an octahedral array. One important class of octahedral clusters are chalcohalide clusters of the type M6X8L6 where M is a metal usually of group 6 or group 7, X is a ligand and more specifically an...
s - AXE method
- Molecular geometryMolecular geometryMolecular geometry or molecular structure is the three-dimensional arrangement of the atoms that constitute a molecule. It determines several properties of a substance including its reactivity, polarity, phase of matter, color, magnetism, and biological activity.- Molecular geometry determination...
- Bailar twistBailar twistThe Bailar twist is a mechanism proposed for the racemization of octahedral complexes containing three bidentate chelate rings. Such complexes typically adopt an octahedral molecular geometry and are thus chiral. One pathway by which these compounds can racemize is via the formation of a trigonal...

