Elbs persulfate oxidation
Encyclopedia
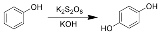
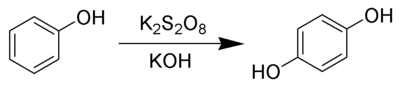
The Elbs persulfate oxidation is the organic reaction
of phenol
s with alkaline potassium persulfate to form para-diphenols.
 Several reviews have been published.
Several reviews have been published.
has been postulated accounting for the observed para substitution featuring the tautomer
ic para carbanion
of the starting phenolate ion:
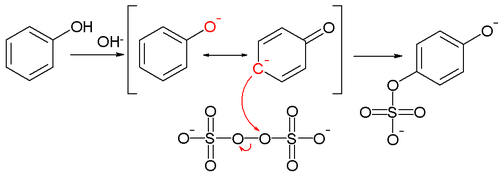 which gives a nucleophilic displacement on the peroxide oxygen of the peroxodisulfate
which gives a nucleophilic displacement on the peroxide oxygen of the peroxodisulfate
(peroxydisulfate) ion. The intermediate sulfate
group is then hydrolyzed to the hydroxyl group.
The reaction is disadvantaged by low chemical yields with recovery of starting material and complete consumption of the persulfate
. It is suggested that the phenol in many cases is a catalyst converting the persulfate into a sulfate.
Organic reaction
Organic reactions are chemical reactions involving organic compounds. The basic organic chemistry reaction types are addition reactions, elimination reactions, substitution reactions, pericyclic reactions, rearrangement reactions, photochemical reactions and redox reactions. In organic synthesis,...
of phenol
Phenol
Phenol, also known as carbolic acid, phenic acid, is an organic compound with the chemical formula C6H5OH. It is a white crystalline solid. The molecule consists of a phenyl , bonded to a hydroxyl group. It is produced on a large scale as a precursor to many materials and useful compounds...
s with alkaline potassium persulfate to form para-diphenols.

Reaction mechanism
A reaction mechanismReaction mechanism
In chemistry, a reaction mechanism is the step by step sequence of elementary reactions by which overall chemical change occurs.Although only the net chemical change is directly observable for most chemical reactions, experiments can often be designed that suggest the possible sequence of steps in...
has been postulated accounting for the observed para substitution featuring the tautomer
Tautomer
Tautomers are isomers of organic compounds that readily interconvert by a chemical reaction called tautomerization. This reaction commonly results in the formal migration of a hydrogen atom or proton, accompanied by a switch of a single bond and adjacent double bond...
ic para carbanion
Carbanion
A carbanion is an anion in which carbon has an unshared pair of electrons and bears a negative charge usually with three substituents for a total of eight valence electrons. The carbanion exists in a trigonal pyramidal geometry. Formally a carbanion is the conjugate base of a carbon acid.where B...
of the starting phenolate ion:

Peroxodisulfate
The peroxodisulfate ion, S2O82−, is a sulfur oxoanion. It is commonly referred to as the persulfate ion, but this term also refers to the peroxomonosulfate ion, SO52−.-Compounds containing peroxodisulfate:* Na2S2O8* K2S2O8* 2S2O8...
(peroxydisulfate) ion. The intermediate sulfate
Sulfate
In inorganic chemistry, a sulfate is a salt of sulfuric acid.-Chemical properties:...
group is then hydrolyzed to the hydroxyl group.
The reaction is disadvantaged by low chemical yields with recovery of starting material and complete consumption of the persulfate
Persulfate
The term persulfate refers to ions or compounds with more oxygen than normal sulfates.These do not have sulfur in a different oxidation state; rather, they contain peroxide units, where two oxygens take the place of one in a normal sulfate; the oxygen atoms are in oxidation state −1.The main forms...
. It is suggested that the phenol in many cases is a catalyst converting the persulfate into a sulfate.

