.gif)
Cyclopentadienylindium(I)
Encyclopedia
Cyclopentadienylindium, C5H5In, is an organoindium compound containing indium
in the +1 oxidation state. Commonly abbreviated to CpIn, it is a cyclopentadienyl complex
with a half-sandwich
structure.
It was the first (1957) low valent organoindium compound reported.
InCp reacts with BF3
, BCl3
, BBr3
, BI3
and trimethylborane
B(CH3)3 to form adducts, e. g.:
In these adducts the bonding of the Cp ligand to the indium atom changes from η
5 (π complexing) to η1
(σ bonding).
Salts containing the InX2− anion containing indium in the +1 oxidation state have been prepared, for example:
Bonding studies have shown that the aromatic ring electrons of the cyclopentadienyl anion interact with the indium 5s and 5p atomic orbital
s, and that the lone pair on the indium atom is a dominant feature.
Indium
Indium is a chemical element with the symbol In and atomic number 49. This rare, very soft, malleable and easily fusible post-transition metal is chemically similar to gallium and thallium, and shows the intermediate properties between these two...
in the +1 oxidation state. Commonly abbreviated to CpIn, it is a cyclopentadienyl complex
Cyclopentadienyl complex
A cyclopentadienyl complex is a metal complex with one or more cyclopentadienyl groups . Based on the type of bonding between the metals and the cyclopentadienyl]] moieties, cyclopentadienyl complexes are classified into the following three categories: a) π-complexes, b) σ-complexes, and c) ionic...
with a half-sandwich
Sandwich compound
In organometallic chemistry, a sandwich compound is a chemical compound featuring a metal bound by haptic covalent bonds to two arene ligands. The arenes have the formula CnHn, substituted derivatives and heterocyclic derivatives...
structure.
It was the first (1957) low valent organoindium compound reported.
Preparation and chemistry
CpIn can be readily prepared by reacting indium(I) chloride with cyclopentadienyllithium:- InCl + CpLi → CpIn + LiCl
InCp reacts with BF3
Boron trifluoride
Boron trifluoride is the chemical compound with the formula BF3. This pungent colourless toxic gas forms white fumes in moist air. It is a useful Lewis acid and a versatile building block for other boron compounds.-Structure and bonding:...
, BCl3
Boron trichloride
Boron trichloride is a chemical compound with the formula BCl3. This colorless gas is a valuable reagent in organic synthesis. It is also dangerously reactive.-Production and properties:Boron reacts with halogens to give the corresponding trihalides...
, BBr3
Boron tribromide
Boron tribromide, BBr3, is a colorless, fuming liquid compound containing boron and bromine. It is usually made by heating boron trioxide with carbon in the presence of bromine: this generates free boron which reacts vigorously with the bromine...
, BI3
Boron triiodide
Boron triiodide is a chemical compound of boron and iodine with chemical formula BI3. It has a trigonal planar molecular geometry. It is a crystalline solid. Its dielectric constant is 5.38 and its heat of vaporization is 40.5 kJ/mol....
and trimethylborane
Trimethylborane
Trimethylborane is a toxic compound normally occurring as a gas that spontaneously catches fire in air. The formula is B3, which can also be expressed as Me3B, with Me representing methyl. Its melting point is -161.5 °C and boiling point is -20.2 °C.Vapour pressure is given by log P =...
B(CH3)3 to form adducts, e. g.:
- CpIn + BF3 → CpIn.BF3
In these adducts the bonding of the Cp ligand to the indium atom changes from η
Hapticity
The term hapticity is used to describe how a group of contiguous atoms of a ligand are coordinated to a central atom. Hapticity of a ligand is indicated by the Greek character 'eta', η. A superscripted number following the η denotes the number of contiguous atoms of the ligand that are bound to...
5 (π complexing) to η1
Hapticity
The term hapticity is used to describe how a group of contiguous atoms of a ligand are coordinated to a central atom. Hapticity of a ligand is indicated by the Greek character 'eta', η. A superscripted number following the η denotes the number of contiguous atoms of the ligand that are bound to...
(σ bonding).
Salts containing the InX2− anion containing indium in the +1 oxidation state have been prepared, for example:
- CpIn + HClHydrochloric acidHydrochloric acid is a solution of hydrogen chloride in water, that is a highly corrosive, strong mineral acid with many industrial uses. It is found naturally in gastric acid....
+ N(Et4)Cl → N(Et4)InCl2+ C5H6CyclopentadieneCyclopentadiene is an organic compound with the formula C5H6. This colorless liquid has a strong and unpleasant odor. At room temperature, this cyclic diene dimerizes over the course of hours to give dicyclopentadiene via a Diels–Alder reaction...
Structure and bonding
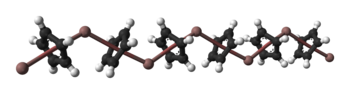
Solid CpIn is polymeric consisting of zigzag chains of alternating indium atoms and C5H5 units. Two indium atoms interact with the opposite faces of each C5H5− ring, nearly perpendicularly to the ring plane, and two rings interact with each indium atom, forming an angle of about 128°. In the CpIn monomer present in the vapour phase the indium atom sits on the central axis of the aromatic cyclopentadienyl anion, C5H5−.Bonding studies have shown that the aromatic ring electrons of the cyclopentadienyl anion interact with the indium 5s and 5p atomic orbital
Atomic orbital
An atomic orbital is a mathematical function that describes the wave-like behavior of either one electron or a pair of electrons in an atom. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus...
s, and that the lone pair on the indium atom is a dominant feature.
 |
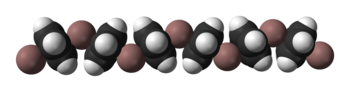 |
Ball-and-stick model In chemistry, the ball-and-stick model is a molecular model of a chemical substance which is to display both the three-dimensional position of the atoms and the bonds between them... and space-filling model Space-filling model In chemistry a space-filling model, also known as calotte model, is a type of three-dimensional molecular model where the atoms are represented by spheres whose radii are proportional to the radii of the atoms and whose center-to-center distances are proportional to the distances between the atomic... s of CpIn chains in the crystal structure of cyclopentadienylindium(I) |

