Chiral auxiliary
Encyclopedia
A chiral auxiliary is a chemical compound
or unit that is temporarily incorporated into an organic synthesis
so that it can be carried out asymmetrically with the selective formation of one of two enantiomer
s
. Chiral auxiliaries are optically active compounds and introduce chirality in otherwise racemic compounds. The temporary stereocenter
then forces the asymmetric formation of a second stereocenter using steric hindrance or directing groups to determine chirality. After the creation of the second stereocenter the original auxiliary can be removed in a third step and recycled
.
Chiral auxiliaries were introduced by E.J. Corey in 1978 with chiral 8-phenylmenthol
and by B.M. Trost in 1980 with chiral mandelic acid
. The menthol compound is difficult to prepare and an alternative is trans-2-phenyl-1-cyclohexanol
introduced by J. K. Whitesell in 1985.
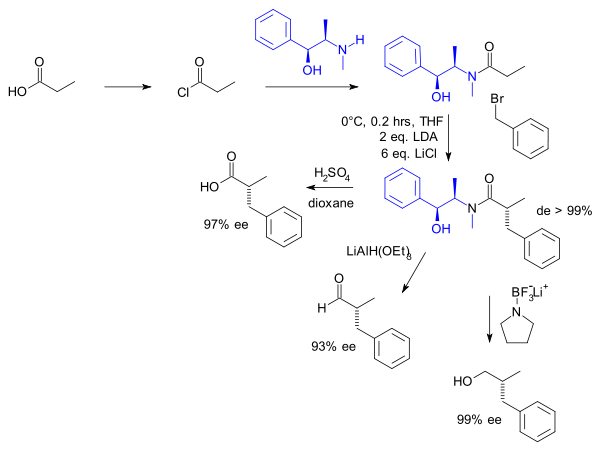
are used as a chiral auxiliary. Pseudoephedrine is reacted with a carboxylic acid
, acid anhydride, or acyl chloride
to give a pseudoephedrine amide.
The α-proton of the carbonyl compound is easily deprotonated by a non-nucleophilic base to give the enolate, which can further react. The configuration of the addition compound, such as with an alkyl halide, is directed by the methyl group. Thus, any addition product will be anti to the methyl and syn with the hydroxyl group. The pseudoephedrine chiral auxiliary is subsequently removed by cleaving the amide bond with an appropriate nucleophile
.
In the original work by Myers (1994) one of the substrates was propionic acid
:
The amide not only can be hydrolysed but also reduced to the aldehyde
(Lithium aluminium hydride
alkoxide) or to the alcohol
(borane
complex with Pyrrolidine
).
In one study the chiral auxiliary is (-)-ephedrine
:
Chemical compound
A chemical compound is a pure chemical substance consisting of two or more different chemical elements that can be separated into simpler substances by chemical reactions. Chemical compounds have a unique and defined chemical structure; they consist of a fixed ratio of atoms that are held together...
or unit that is temporarily incorporated into an organic synthesis
Organic synthesis
Organic synthesis is a special branch of chemical synthesis and is concerned with the construction of organic compounds via organic reactions. Organic molecules can often contain a higher level of complexity compared to purely inorganic compounds, so the synthesis of organic compounds has...
so that it can be carried out asymmetrically with the selective formation of one of two enantiomer
Enantiomer
In chemistry, an enantiomer is one of two stereoisomers that are mirror images of each other that are non-superposable , much as one's left and right hands are the same except for opposite orientation. It can be clearly understood if you try to place your hands one over the other without...
s
. Chiral auxiliaries are optically active compounds and introduce chirality in otherwise racemic compounds. The temporary stereocenter
Stereocenter
A stereocenter or stereogenic center is an atom, bearing groups such that an interchanging of any two groups leads to a stereoisomer.A chirality center is a stereocenter consisting of an atom holding a set of ligands in a spatial arrangement which is not superposable on its mirror image...
then forces the asymmetric formation of a second stereocenter using steric hindrance or directing groups to determine chirality. After the creation of the second stereocenter the original auxiliary can be removed in a third step and recycled
Recycling
Recycling is processing used materials into new products to prevent waste of potentially useful materials, reduce the consumption of fresh raw materials, reduce energy usage, reduce air pollution and water pollution by reducing the need for "conventional" waste disposal, and lower greenhouse...
.
Chiral auxiliaries were introduced by E.J. Corey in 1978 with chiral 8-phenylmenthol
Menthol
Menthol is an organic compound made synthetically or obtained from peppermint or other mint oils. It is a waxy, crystalline substance, clear or white in color, which is solid at room temperature and melts slightly above. The main form of menthol occurring in nature is -menthol, which is assigned...
and by B.M. Trost in 1980 with chiral mandelic acid
Mandelic acid
Mandelic acid is an aromatic alpha hydroxy acid with the molecular formula C6H5CHCO2H. It is a white crystalline solid that is soluble in water and polar organic solvents. It is a useful precursor to various drugs...
. The menthol compound is difficult to prepare and an alternative is trans-2-phenyl-1-cyclohexanol
Trans-2-Phenyl-1-cyclohexanol
trans-2-Phenyl-1-cyclohexanol is an organic compound. The two enantiomers of this compound are used in organic chemistry as chiral auxiliaries.-Preparation:...
introduced by J. K. Whitesell in 1985.
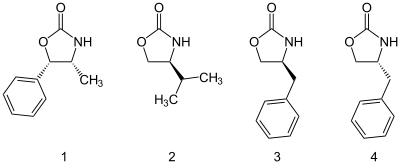
Oxazolidone auxiliaries
A good example of chiral auxiliaries are the class of Evans auxiliaries comprising oxazolidinones substituted at the 4 and 5 positions. Through steric hindrance, the substituents direct the direction of substitution of various groups. The auxiliary is subsequently removed e.g. through hydrolysis.
Pseudoephedrine auxiliaries
Both (R,R)- and (S,S)-pseudoephedrinePseudoephedrine
Pseudoephedrine is a sympathomimetic drug of the phenethylamine and amphetamine chemical classes. It is used as a nasal/sinus decongestant and stimulant, or as a wakefulness-promoting agent....
are used as a chiral auxiliary. Pseudoephedrine is reacted with a carboxylic acid
Carboxylic acid
Carboxylic acids are organic acids characterized by the presence of at least one carboxyl group. The general formula of a carboxylic acid is R-COOH, where R is some monovalent functional group...
, acid anhydride, or acyl chloride
Acyl chloride
In organic chemistry, an acyl chloride is an organic compound with the functional group -CO-Cl. Their formula is usually written RCOCl, where R is a side chain. They are usually considered to be reactive derivatives of carboxylic acids. A specific example of an acyl chloride is acetyl chloride,...
to give a pseudoephedrine amide.
The α-proton of the carbonyl compound is easily deprotonated by a non-nucleophilic base to give the enolate, which can further react. The configuration of the addition compound, such as with an alkyl halide, is directed by the methyl group. Thus, any addition product will be anti to the methyl and syn with the hydroxyl group. The pseudoephedrine chiral auxiliary is subsequently removed by cleaving the amide bond with an appropriate nucleophile
Nucleophile
A nucleophile is a species that donates an electron-pair to an electrophile to form a chemical bond in a reaction. All molecules or ions with a free pair of electrons can act as nucleophiles. Because nucleophiles donate electrons, they are by definition Lewis bases.Nucleophilic describes the...
.
In the original work by Myers (1994) one of the substrates was propionic acid
Propionic acid
Propanoic acid is a naturally occurring carboxylic acid with chemical formula CH3CH2COOH. It is a clear liquid with a pungent odor...
:
The amide not only can be hydrolysed but also reduced to the aldehyde
Aldehyde
An aldehyde is an organic compound containing a formyl group. This functional group, with the structure R-CHO, consists of a carbonyl center bonded to hydrogen and an R group....
(Lithium aluminium hydride
Lithium aluminium hydride
Lithium aluminium hydride, commonly abbreviated to LAH or known as LithAl, is an inorganic compound with the chemical formula LiAlH4. It was discovered by Finholt, Bond and Schlesinger in 1947. This compound is used as a reducing agent in organic synthesis, especially for the reduction of esters,...
alkoxide) or to the alcohol
Alcohol
In chemistry, an alcohol is an organic compound in which the hydroxy functional group is bound to a carbon atom. In particular, this carbon center should be saturated, having single bonds to three other atoms....
(borane
Borane
In chemistry, a borane is a chemical compound of boron and hydrogen. The boranes comprise a large group of compounds with the generic formulae of BxHy. These compounds do not occur in nature. Many of the boranes readily oxidise on contact with air, some violently. The parent member BH3 is called...
complex with Pyrrolidine
Pyrrolidine
Pyrrolidine, also known as tetrahydropyrrole, is an organic compound with the molecular formula C4H9N. It is a cyclic secondary amine with a five-membered heterocycle containing four carbon atoms and one nitrogen atom...
).
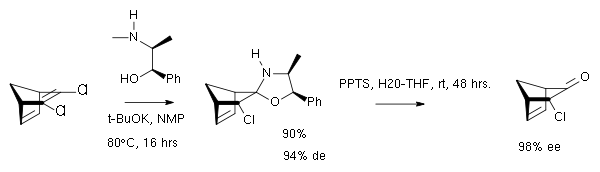
In one study the chiral auxiliary is (-)-ephedrine
Ephedrine
Ephedrine is a sympathomimetic amine commonly used as a stimulant, appetite suppressant, concentration aid, decongestant, and to treat hypotension associated with anaesthesia....
:
See also
- Example of use of trans-2-Phenyl-1-cyclohexanolTrans-2-Phenyl-1-cyclohexanoltrans-2-Phenyl-1-cyclohexanol is an organic compound. The two enantiomers of this compound are used in organic chemistry as chiral auxiliaries.-Preparation:...
as chiral auxiliary: Ojima lactamOjima lactamThe Ojima lactam is an organic compound of some importance in the commercial production of Taxol. This lactam was first synthesized by Iwao Ojima . The organic synthesis is an illustration of asymmetric synthesis via a chiral auxiliary.... - Valine as a Chiral auxiliary in the Schöllkopf methodSchöllkopf methodThe Schöllkopf method or Schöllkopf Bis-Lactim Amino Acid Synthesis is a method in organic chemistry for the asymmetric synthesis of chiral amino acids. The method was established in 1981 by Ulrich Schöllkopf...
- Tert-butanesulfinamideTert-Butanesulfinamidetert-Butanesulfinamide is an organosulfur compound and a member of the class of sulfinamides . Both enantiomeric forms are commercially available and are relevant to the asymmetric synthesis of amines as a chiral ammonia equivalent. This methodology was introduced in 1997 by Jonathan A...
as reagent for asymmetric synthesis of amines