
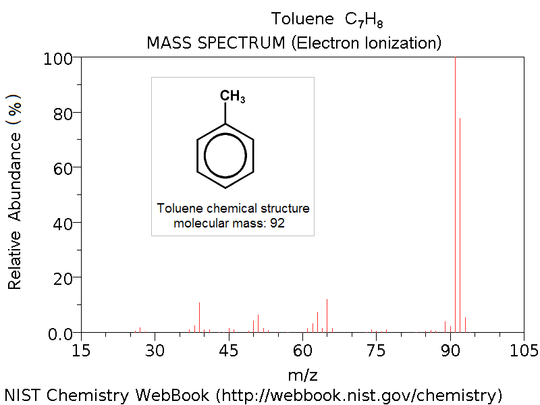
Mass spectrum
Encyclopedia
Mass-to-charge ratio
The mass-to-charge ratio ratio is a physical quantity that is widely used in the electrodynamics of charged particles, e.g. in electron optics and ion optics. It appears in the scientific fields of lithography, electron microscopy, cathode ray tubes, accelerator physics, nuclear physics, Auger...
) plot representing a chemical analysis. Hence, the mass spectrum of a sample is a pattern representing the distribution of ions by mass
Mass
Mass can be defined as a quantitive measure of the resistance an object has to change in its velocity.In physics, mass commonly refers to any of the following three properties of matter, which have been shown experimentally to be equivalent:...
(more correctly: mass-to-charge ratio
Mass-to-charge ratio
The mass-to-charge ratio ratio is a physical quantity that is widely used in the electrodynamics of charged particles, e.g. in electron optics and ion optics. It appears in the scientific fields of lithography, electron microscopy, cathode ray tubes, accelerator physics, nuclear physics, Auger...
) in a sample. It is a histogram
Histogram
In statistics, a histogram is a graphical representation showing a visual impression of the distribution of data. It is an estimate of the probability distribution of a continuous variable and was first introduced by Karl Pearson...
usually acquired using an instrument called a mass spectrometer. Not all mass spectra of a given substance are the same. For example some mass spectrometers break the analyte molecules into fragments; others observe the intact molecular masses with little fragmentation. A mass spectrum can represent many different types of information based on the type of mass spectrometer and the specific experiment applied; however, all plots of intensity vs. mass-to-charge are referred to as mass spectra. Common fragmentation processes for organic molecules are the McLafferty rearrangement
McLafferty rearrangement
The McLafferty rearrangement is a reaction observed in mass spectrometry. It is sometimes found that a molecule containing a keto-group undergoes β-cleavage, with the gain of the γ-hydrogen atom...
and alpha cleavage
Alpha cleavage
Alpha cleavage, ' in organic chemistry, refers to the act of breaking the carbon-carbon bond, adjacent to the carbon bearing a specified functional group.-Mass spectrometry:...
.
X-axis: m/z (mass-to-charge ratio)
The x-axis of a mass spectrum represents a relationship between the mass of a given ion and the number of elementary charges that it carries. This is written as the IUPAC standard m/z to denote the quantity formed by dividing the mass of an ion by the unified atomic mass unit and by its charge number (positive absolute value). This has been referred to as a mass-to-charge ratioMass-to-charge ratio
The mass-to-charge ratio ratio is a physical quantity that is widely used in the electrodynamics of charged particles, e.g. in electron optics and ion optics. It appears in the scientific fields of lithography, electron microscopy, cathode ray tubes, accelerator physics, nuclear physics, Auger...
, although in some ways it does not fit this description. The IUPAC Gold Book gives an example: "for the ion C7H72+, m/z equals 45.5".
Since a mass spectrum x-axis represents a relationship between the ion mass and the number of elementary charges that a given ion carries it contains mass information that may be extracted by a knowledgeable mass spectrometrist. Once this is done many mass spectrometrists use dalton (Da) as the unit of mass in order to avoid the clumsy "atomic mass units".
Alternative x-axis notations
There are several alternatives to the standard m/z notation that appear in the literature; however, these are not currently accepted by standards organizations and most journals. m/e appears in older historical literature. A label more consistent with the IUPAC green bookIUPAC green book
right|thumb|Front cover of the Green BookQuantities, Units and Symbols in Physical Chemistry Third Edition , also known as the Green Book, published in Aug 2007 by IUPAC is based on the most up to date sources for fundamental constants, data and nomenclature in the field of chemistry and physics...
and ISO 31
ISO 31
International Standard ISO 31 was the most widely respected style guide for the use of physical quantities and units of measurement, and formulas involving them, in scientific and educational documents worldwide...
conventions is m/Q or m/q where m is the symbol for mass and Q or q the symbol for charge with the units u/e or Da/e. This notation is not uncommon in the physics of mass spectrometry but is rarely used as the abscissa of a mass spectrum. It was also suggested to introduce a new unit thomson
Thomson (unit)
The thomson is a unit that has appeared infrequently in scientific literature relating to the field of mass spectrometry as a unit of mass-to-charge ratio. The unit was proposed by Cooks and Rockwood naming it in honour of J. J...
(Th) as a unit of m/z, where 1 Th = 1 u/e. According to this convention, mass spectra x axis could be labeled m/z (Th) and negative ions would have negative values. This notation is rare and not accepted by IUPAC or any other standards organisation.
History of x-axis notation
In 1897 the mass-to-charge ratio of the electron
of the electronElectron
The electron is a subatomic particle with a negative elementary electric charge. It has no known components or substructure; in other words, it is generally thought to be an elementary particle. An electron has a mass that is approximately 1/1836 that of the proton...
was first measured by J. J. Thomson
J. J. Thomson
Sir Joseph John "J. J." Thomson, OM, FRS was a British physicist and Nobel laureate. He is credited for the discovery of the electron and of isotopes, and the invention of the mass spectrometer...
.. By doing this he showed that the electron, which was postulated before in order to explain electricity, was in fact a particle with a mass and a charge and that its mass-to-charge ratio was much smaller than the one for the hydrogen ion H+. In 1913 he measured the mass-to-charge ratio of ion
Ion
An ion is an atom or molecule in which the total number of electrons is not equal to the total number of protons, giving it a net positive or negative electrical charge. The name was given by physicist Michael Faraday for the substances that allow a current to pass between electrodes in a...
s with an instrument he called a parabola spectrograph. Although this data was not represented as a modern mass spectrum, it was similar in meaning. Eventually there was a change to the notation as m/e giving way to the current standard of m/z.
Early in mass spectrometry research the resolution
Resolution (mass spectrometry)
In mass spectrometry, resolution measures of the ability to distinguish two peaks of slightly different mass-to-charge ratios ΔM, in a mass spectrum.- Resolution and Resolving Power :...
of mass spectrometers did not allow for accurate mass determination. Francis William Aston
Francis William Aston
Francis William Aston was a British chemist and physicist who won the 1922 Nobel Prize in Chemistry for his discovery, by means of his mass spectrograph, of isotopes, in a large number of non-radioactive elements, and for his enunciation of the whole-number rule...
won the Nobel prize in Chemistry in 1922. "For his discovery, by means of his mass spectrograph, of isotopes, in a large number of non-radioactive elements, and for his enunciation of the Whole Number Rule
Whole number rule
The whole number rule states that the masses of the elements are whole number multiples of the mass of the hydrogen atom. The rule can be formulated from Prout's hypothesis put forth in 1815. In 1920, Francis W...
." In which he stated that all atoms (including isotopes) follow a whole-number rule This implied that the masses of atoms were not on a scale but could be expressed as integers. (In fact multiply charged ions were rare, so for the most part the ratio was whole as well.) There have been several suggestions (e.g. the unit thomson) to change the official mass spectrometry nomenclature
 to be more internally consistent.
to be more internally consistent.Y-axis: signal intensity
The y-axis of a mass spectrum represents signal intensity of the ions. When using counting detectors the intensity is often measured in counts per second (cps). When using analog detection electronics the intensity is typically measured in volts. In FTICR and OrbitrapOrbitrap
An orbitrap is a type of mass spectrometer invented by Alexander Makarov. It consists of an outer barrel-like electrode and a coaxial inner spindle-like electrode that form an electrostatic field with quadro-logarithmic potential distribution....
s the frequency domain
Frequency domain
In electronics, control systems engineering, and statistics, frequency domain is a term used to describe the domain for analysis of mathematical functions or signals with respect to frequency, rather than time....
signal (the y-axis) is related to the power
Power (physics)
In physics, power is the rate at which energy is transferred, used, or transformed. For example, the rate at which a light bulb transforms electrical energy into heat and light is measured in watts—the more wattage, the more power, or equivalently the more electrical energy is used per unit...
(~amplitude squared) of the signal sine wave
Sine wave
The sine wave or sinusoid is a mathematical function that describes a smooth repetitive oscillation. It occurs often in pure mathematics, as well as physics, signal processing, electrical engineering and many other fields...
(often reduced to an rms power); however, the axis is usually not labeled as such for many reasons. In most forms of mass spectrometry, the intensity of ion current measured by the spectrometer does not accurately represent relative abundance, but correlates loosely with it. Therefore it is common to label the y-axis with "arbitrary units".
Y-axis and relative abundance
Signal intensity may be dependent on many factors, especially the nature of the molecules being analyzed and how they ionize. The efficiency of ionization varies from molecule to molecule and from ion source to ion source. For example, in electrospray sources in positive ion mode a quaternary amine will ionize exceptionally well whereas a large hydrophobic alcohol will most likely not be seen no matter how concentrated. In an EI source these molecules will behave very differently. Additionally there may be factors that affect ion transmission disproportionally between ionization and detection.On the detection side there are many factors that can also affect signal intensity in a non-proportional way. The size of the ion will affect the velocity of impact and with certain detectors the velocity is proportional to the signal output. In other detection systems, such as FTICR, the number of charges on the ion are more important to signal intensity. In Fourier transform ion cyclotron resonance
Fourier transform ion cyclotron resonance
Fourier transform ion cyclotron resonance mass spectrometry, also known as Fourier transform mass spectrometry, is a type of mass analyzer for determining the mass-to-charge ratio of ions based on the cyclotron frequency of the ions in a fixed magnetic field...
and Orbitrap
Orbitrap
An orbitrap is a type of mass spectrometer invented by Alexander Makarov. It consists of an outer barrel-like electrode and a coaxial inner spindle-like electrode that form an electrostatic field with quadro-logarithmic potential distribution....
type mass spectrometers the signal intensity (Y-axis) is related to the amplitude of the free induction decay
Free induction decay
In Fourier Transform NMR, free induction decay is the observable NMR signal generated by non-equilibrium nuclear spin magnetisation precessing about the magnetic field ....
signal. This is fundamentally a power relationship (amplitude squared) but often computed as an [rms]. For decaying signals the rms is not equal to the average amplitude. Additionally the damping constant (decay rate of the signal in the fid) is not the same for all ions. In order to make conclusions about relative intensity a great deal of knowledge and care is required.
A common way to get more quantitative information out of a mass spectrum is to create a standard curve to compare the sample to. This requires knowing what is to be quantitated ahead of time, having a standard available and designing the experiment specifically for this purpose. A more advanced variation on this the use of an internal standard
Internal standard
An internal standard in analytical chemistry is a chemical substance that is added in a constant amount to samples, the blank and calibration standards in a chemical analysis. This substance can then be used for calibration by plotting the ratio of the analyte signal to the internal standard signal...
which behaves very similarly to the analyte. This is often an isotopically labeled version of the analyte. There are forms of mass spectrometry, such as accelerator mass spectrometry
Accelerator mass spectrometry
Accelerator mass spectrometry differs from other forms of mass spectrometry in that it accelerates ions to extraordinarily high kinetic energies before mass analysis. The special strength of AMS among the mass spectrometric methods is its power to separate a rare isotope from an abundant...
that are designed from the bottom up to be quantitative.
External links
- Interactive tutorial on mass spectra National High Magnetic Field Laboratory
- Weighing Atoms: A layperson's introduction to mass spectrometry National High Magnetic Field Laboratory
- What's in an Oil Drop: A layperson's introduction to Fourier Transform Ion Cyclotron Resonance National High Magnetic Field Laboratory
- Quantities, Units and Symbols in Physical Chemistry (IUPAC green book)
- An introductory video on Mass Spectrometry The Royal Society of Chemistry

