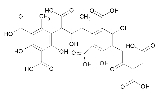
Humic acid
Overview
Organic compound
An organic compound is any member of a large class of gaseous, liquid, or solid chemical compounds whose molecules contain carbon. For historical reasons discussed below, a few types of carbon-containing compounds such as carbides, carbonates, simple oxides of carbon, and cyanides, as well as the...
constituents of soil (humus), peat
Peat
Peat is an accumulation of partially decayed vegetation matter or histosol. Peat forms in wetland bogs, moors, muskegs, pocosins, mires, and peat swamp forests. Peat is harvested as an important source of fuel in certain parts of the world...
, coal
Coal
Coal is a combustible black or brownish-black sedimentary rock usually occurring in rock strata in layers or veins called coal beds or coal seams. The harder forms, such as anthracite coal, can be regarded as metamorphic rock because of later exposure to elevated temperature and pressure...
, many upland streams
River
A river is a natural watercourse, usually freshwater, flowing towards an ocean, a lake, a sea, or another river. In a few cases, a river simply flows into the ground or dries up completely before reaching another body of water. Small rivers may also be called by several other names, including...
, dystrophic lake
Dystrophic lake
Dystrophic lake refers to lakes with brown- or tea-coloured waters, the colour being the result of high concentrations of humic substances and organic acids suspended in the water. Because the term has long been misused in the literature, these lakes are better referred to as humic lakes...
s, and ocean
Ocean
An ocean is a major body of saline water, and a principal component of the hydrosphere. Approximately 71% of the Earth's surface is covered by ocean, a continuous body of water that is customarily divided into several principal oceans and smaller seas.More than half of this area is over 3,000...
water
Water
Water is a chemical substance with the chemical formula H2O. A water molecule contains one oxygen and two hydrogen atoms connected by covalent bonds. Water is a liquid at ambient conditions, but it often co-exists on Earth with its solid state, ice, and gaseous state . Water also exists in a...
. It is produced by biodegradation of dead organic matter. It is not a single acid
Acid
An acid is a substance which reacts with a base. Commonly, acids can be identified as tasting sour, reacting with metals such as calcium, and bases like sodium carbonate. Aqueous acids have a pH of less than 7, where an acid of lower pH is typically stronger, and turn blue litmus paper red...
; rather, it is a complex mixture of many different acids containing carboxyl and phenol
Phenol
Phenol, also known as carbolic acid, phenic acid, is an organic compound with the chemical formula C6H5OH. It is a white crystalline solid. The molecule consists of a phenyl , bonded to a hydroxyl group. It is produced on a large scale as a precursor to many materials and useful compounds...
ate groups so that the mixture behaves functionally as a dibasic acid
Dibasic acid
A dibasic acid is an acid that has two hydrogen ions to donate to a base in an acid-base reaction. Therefore, a dibasic molecule has two replaceable hydrogen atoms. Example: H2SO4....
or, occasionally, as a tribasic acid
Tribasic acid
A tribasic acid is an acid that has three hydrogen ions to donate to a base in an acid-base reaction. Therefore, a tribasic molecule has three replaceable hydrogen atoms.Phosphoric acid and citric acid are examples of tribasic acids....
.
Unanswered Questions

