
Formaldehyde dehydrogenase
Encyclopedia
In enzymology, a formaldehyde dehydrogenase is an enzyme
that catalyzes
the chemical reaction
The 3 substrates
of this enzyme are formaldehyde
, NAD+
, and H2O
, whereas its 3 products
are formate
, NADH
, and H+
.
This enzyme belongs to the family of oxidoreductase
s, specifically those acting on the aldehyde or oxo group of donor with NAD+ or NADP+ as acceptor. The systematic name of this enzyme class is formaldehyde:NAD+ oxidoreductase. Other names in common use include NAD+-linked formaldehyde dehydrogenase, s-nitrosoglutathione reductase (GSNO reductase) and NAD+-dependent formaldehyde dehydrogenase. This enzyme participates in methane metabolism.
(GSNO) to the unstable intermediate, S-hydroxylaminoglutathione, which then rearranges to form glutathione sulfinamide, or in the presence of GSH, forms oxidized glutathione (GSSG) and hydroxyl amine. Through this catabolic process, GSNOR regulates the cellular concentrations of GSNO and plays a central role in regulating the levels of endogenous S-nitrosothiols and controlling protein S-nitrosylation-based signaling. As an example of S-nitrosylation-based signaling, Barglow et al showed that GSNO selectively S-nitrosylates reduced thioredoxin at cysteine 62. Nitrosylated thioredoxin, via directed protein-protein interaction, trans-nitrosylates the active site cysteine of caspase-3 thus inactivating caspase-3 and preventing induction of apoptosis.
As might be expected of an enzyme involved in regulating NO levels and signaling, pleiotropic effects are observed in GSNOR knockout models. Deleting the GSNOR gene from both yeast and mice increased the cellular levels of GSNO and nitrosylated proteins, and the yeast cells showed increased susceptibility to nitrosative stress. Null mice show increased levels of S-nitrosated proteins, increased beta adrenergic receptor numbers in lung and heart, diminished tachyphylaxis to β2-adrenergic receptor agonists, hyporesponsiveness to methacholine and allergen challenge and reduced infarct size after occlusion of the coronary artery. In addition, null mice show increased tissue damage and mortality following challenge with bacteria or endotoxin and are hypotensive under anesthesia yet normotensive in the conscious state. More related to its alcohol dehydrogenase activity, GSNOR null mice show a 30% reduction in the LD50 for formaldehyde and a decreased capacity to metabolize retinol, although it is clear from these studies that other pathways exist for the metabolism of these compounds.
(SNO) levels in the alveolar lining fluid in the lung and with responsiveness to methacholine challenge in patients with mild asthma compared to normal subjects. Furthermore, there are lowered SNOs in tracheal irrigations in asthmatic children with respiratory failure in comparison to normal children undergoing elective surgery and NO species are elevated in asthma patients when exposed to antigen.
Assessing the gene expression of the ADHs in nonalcoholic steatohepatitis (NASH) patients has shown elevated levels of all ADHs, but primarily ADH1 and ADH4 (up to 40-fold increased). ADH5 showed an ~4-fold increase in gene expression.
has been solved for this class of enzymes, with the PDB
accession code .
Enzyme
Enzymes are proteins that catalyze chemical reactions. In enzymatic reactions, the molecules at the beginning of the process, called substrates, are converted into different molecules, called products. Almost all chemical reactions in a biological cell need enzymes in order to occur at rates...
that catalyzes
Catalysis
Catalysis is the change in rate of a chemical reaction due to the participation of a substance called a catalyst. Unlike other reagents that participate in the chemical reaction, a catalyst is not consumed by the reaction itself. A catalyst may participate in multiple chemical transformations....
the chemical reaction
Chemical reaction
A chemical reaction is a process that leads to the transformation of one set of chemical substances to another. Chemical reactions can be either spontaneous, requiring no input of energy, or non-spontaneous, typically following the input of some type of energy, such as heat, light or electricity...
- formaldehyde + NAD+ + H2O
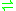 formate + NADH + 2 H+
formate + NADH + 2 H+
The 3 substrates
Substrate (biochemistry)
In biochemistry, a substrate is a molecule upon which an enzyme acts. Enzymes catalyze chemical reactions involving the substrate. In the case of a single substrate, the substrate binds with the enzyme active site, and an enzyme-substrate complex is formed. The substrate is transformed into one or...
of this enzyme are formaldehyde
Formaldehyde
Formaldehyde is an organic compound with the formula CH2O. It is the simplest aldehyde, hence its systematic name methanal.Formaldehyde is a colorless gas with a characteristic pungent odor. It is an important precursor to many other chemical compounds, especially for polymers...
, NAD+
Nicotinamide adenine dinucleotide
Nicotinamide adenine dinucleotide, abbreviated NAD, is a coenzyme found in all living cells. The compound is a dinucleotide, since it consists of two nucleotides joined through their phosphate groups. One nucleotide contains an adenine base and the other nicotinamide.In metabolism, NAD is involved...
, and H2O
Water
Water is a chemical substance with the chemical formula H2O. A water molecule contains one oxygen and two hydrogen atoms connected by covalent bonds. Water is a liquid at ambient conditions, but it often co-exists on Earth with its solid state, ice, and gaseous state . Water also exists in a...
, whereas its 3 products
Product (chemistry)
Product are formed during chemical reactions as reagents are consumed. Products have lower energy than the reagents and are produced during the reaction according to the second law of thermodynamics. The released energy comes from changes in chemical bonds between atoms in reagent molecules and...
are formate
Formate
Formate or methanoate is the ion CHOO− or HCOO− . It is the simplest carboxylate anion. It is produced in large amounts in the hepatic mitochondria of embryonic cells and in cancer cells by the folate cycle Formate or methanoate is the ion CHOO− or HCOO− (formic acid minus one hydrogen ion). It...
, NADH
Nicotinamide adenine dinucleotide
Nicotinamide adenine dinucleotide, abbreviated NAD, is a coenzyme found in all living cells. The compound is a dinucleotide, since it consists of two nucleotides joined through their phosphate groups. One nucleotide contains an adenine base and the other nicotinamide.In metabolism, NAD is involved...
, and H+
Hydrogen ion
Hydrogen ion is recommended by IUPAC as a general term for all ions of hydrogen and its isotopes.Depending on the charge of the ion, two different classes can be distinguished: positively charged ions and negatively charged ions....
.
This enzyme belongs to the family of oxidoreductase
Oxidoreductase
In biochemistry, an oxidoreductase is an enzyme that catalyzes the transfer of electrons from one molecule to another...
s, specifically those acting on the aldehyde or oxo group of donor with NAD+ or NADP+ as acceptor. The systematic name of this enzyme class is formaldehyde:NAD+ oxidoreductase. Other names in common use include NAD+-linked formaldehyde dehydrogenase, s-nitrosoglutathione reductase (GSNO reductase) and NAD+-dependent formaldehyde dehydrogenase. This enzyme participates in methane metabolism.
Ubiquitous function
S-nitrosoglutathione reductase (GSNOR) is a class III alcohol dehydrogenase (ADH) encoded by the ADH5 gene in humans. It is a primordial ADH that is ubiquitously expressed in plant and animals alike. GSNOR reduces S-nitrosoglutathioneS-Nitrosoglutathione
S-Nitrosoglutathione is an endogenous S-nitrosothiol that plays a critical role in nitric oxide signaling and is a source of bioavailable NO. NO coexists in cells with SNOs that serve as endogenous NO carriers and donors...
(GSNO) to the unstable intermediate, S-hydroxylaminoglutathione, which then rearranges to form glutathione sulfinamide, or in the presence of GSH, forms oxidized glutathione (GSSG) and hydroxyl amine. Through this catabolic process, GSNOR regulates the cellular concentrations of GSNO and plays a central role in regulating the levels of endogenous S-nitrosothiols and controlling protein S-nitrosylation-based signaling. As an example of S-nitrosylation-based signaling, Barglow et al showed that GSNO selectively S-nitrosylates reduced thioredoxin at cysteine 62. Nitrosylated thioredoxin, via directed protein-protein interaction, trans-nitrosylates the active site cysteine of caspase-3 thus inactivating caspase-3 and preventing induction of apoptosis.
As might be expected of an enzyme involved in regulating NO levels and signaling, pleiotropic effects are observed in GSNOR knockout models. Deleting the GSNOR gene from both yeast and mice increased the cellular levels of GSNO and nitrosylated proteins, and the yeast cells showed increased susceptibility to nitrosative stress. Null mice show increased levels of S-nitrosated proteins, increased beta adrenergic receptor numbers in lung and heart, diminished tachyphylaxis to β2-adrenergic receptor agonists, hyporesponsiveness to methacholine and allergen challenge and reduced infarct size after occlusion of the coronary artery. In addition, null mice show increased tissue damage and mortality following challenge with bacteria or endotoxin and are hypotensive under anesthesia yet normotensive in the conscious state. More related to its alcohol dehydrogenase activity, GSNOR null mice show a 30% reduction in the LD50 for formaldehyde and a decreased capacity to metabolize retinol, although it is clear from these studies that other pathways exist for the metabolism of these compounds.
Role in disease
It has been shown that GSNOR may have an important role in respiratory diseases such as asthma. GSNOR expression has been inversely correlated with s-nitrosothiolS-Nitrosothiol
S-Nitrosothiols, also known as thionitrites, are organic compounds or functional groups containing a nitroso group attached to the sulfur atom of a thiol...
(SNO) levels in the alveolar lining fluid in the lung and with responsiveness to methacholine challenge in patients with mild asthma compared to normal subjects. Furthermore, there are lowered SNOs in tracheal irrigations in asthmatic children with respiratory failure in comparison to normal children undergoing elective surgery and NO species are elevated in asthma patients when exposed to antigen.
Assessing the gene expression of the ADHs in nonalcoholic steatohepatitis (NASH) patients has shown elevated levels of all ADHs, but primarily ADH1 and ADH4 (up to 40-fold increased). ADH5 showed an ~4-fold increase in gene expression.
Structural studies
As of late 2007, only one structureTertiary structure
In biochemistry and molecular biology, the tertiary structure of a protein or any other macromolecule is its three-dimensional structure, as defined by the atomic coordinates.-Relationship to primary structure:...
has been solved for this class of enzymes, with the PDB
Protein Data Bank
The Protein Data Bank is a repository for the 3-D structural data of large biological molecules, such as proteins and nucleic acids....
accession code .

