
Ferredoxin hydrogenase
Encyclopedia
In enzymology, a ferredoxin hydrogenase is an enzyme
that catalyzes
the chemical reaction
Thus, the two substrates
of this enzyme are H2
and oxidized ferredoxin, whereas its two products
are reduced ferredoxin and H+
.
This enzyme belongs to the family of oxidoreductase
s, specifically those acting on hydrogen as donor with an iron-sulfur protein as acceptor. The systematic name of this enzyme class is hydrogen:ferredoxin oxidoreductase. Other names in common use include H2 oxidizing hydrogenase, H2 producing hydrogenase [ambiguous], bidirectional hydrogenase, hydrogen-lyase [ambiguous], hydrogenase (ferredoxin), hydrogenase I, hydrogenase II, hydrogenlyase [ambiguous], and uptake hydrogenase [ambiguous]. This enzyme participates in glyoxylate and dicarboxylate metabolism
and methane metabolism. It has 3 cofactors
: iron
, Sulfur
, and Nickel
.
Enzyme
Enzymes are proteins that catalyze chemical reactions. In enzymatic reactions, the molecules at the beginning of the process, called substrates, are converted into different molecules, called products. Almost all chemical reactions in a biological cell need enzymes in order to occur at rates...
that catalyzes
Catalysis
Catalysis is the change in rate of a chemical reaction due to the participation of a substance called a catalyst. Unlike other reagents that participate in the chemical reaction, a catalyst is not consumed by the reaction itself. A catalyst may participate in multiple chemical transformations....
the chemical reaction
Chemical reaction
A chemical reaction is a process that leads to the transformation of one set of chemical substances to another. Chemical reactions can be either spontaneous, requiring no input of energy, or non-spontaneous, typically following the input of some type of energy, such as heat, light or electricity...
- H2 + 2 oxidized ferredoxin
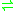 2 reduced ferredoxin + 2 H+
2 reduced ferredoxin + 2 H+
Thus, the two substrates
Substrate (biochemistry)
In biochemistry, a substrate is a molecule upon which an enzyme acts. Enzymes catalyze chemical reactions involving the substrate. In the case of a single substrate, the substrate binds with the enzyme active site, and an enzyme-substrate complex is formed. The substrate is transformed into one or...
of this enzyme are H2
Hydrogen
Hydrogen is the chemical element with atomic number 1. It is represented by the symbol H. With an average atomic weight of , hydrogen is the lightest and most abundant chemical element, constituting roughly 75% of the Universe's chemical elemental mass. Stars in the main sequence are mainly...
and oxidized ferredoxin, whereas its two products
Product (chemistry)
Product are formed during chemical reactions as reagents are consumed. Products have lower energy than the reagents and are produced during the reaction according to the second law of thermodynamics. The released energy comes from changes in chemical bonds between atoms in reagent molecules and...
are reduced ferredoxin and H+
Hydrogen ion
Hydrogen ion is recommended by IUPAC as a general term for all ions of hydrogen and its isotopes.Depending on the charge of the ion, two different classes can be distinguished: positively charged ions and negatively charged ions....
.
This enzyme belongs to the family of oxidoreductase
Oxidoreductase
In biochemistry, an oxidoreductase is an enzyme that catalyzes the transfer of electrons from one molecule to another...
s, specifically those acting on hydrogen as donor with an iron-sulfur protein as acceptor. The systematic name of this enzyme class is hydrogen:ferredoxin oxidoreductase. Other names in common use include H2 oxidizing hydrogenase, H2 producing hydrogenase [ambiguous], bidirectional hydrogenase, hydrogen-lyase [ambiguous], hydrogenase (ferredoxin), hydrogenase I, hydrogenase II, hydrogenlyase [ambiguous], and uptake hydrogenase [ambiguous]. This enzyme participates in glyoxylate and dicarboxylate metabolism
Glyoxylate and dicarboxylate metabolism
Glyoxylate and dicarboxylate metabolism describes a variety of reactions involving glyoxylate or dicarboxylates. Glyoxylate is the conjugate base of glyoxylic acid, and within a buffered environment of known pH such as the cell cytoplasm these terms can be used almost interchangeably, as the gain...
and methane metabolism. It has 3 cofactors
Cofactor (biochemistry)
A cofactor is a non-protein chemical compound that is bound to a protein and is required for the protein's biological activity. These proteins are commonly enzymes, and cofactors can be considered "helper molecules" that assist in biochemical transformations....
: iron
Iron
Iron is a chemical element with the symbol Fe and atomic number 26. It is a metal in the first transition series. It is the most common element forming the planet Earth as a whole, forming much of Earth's outer and inner core. It is the fourth most common element in the Earth's crust...
, Sulfur
Sulfur
Sulfur or sulphur is the chemical element with atomic number 16. In the periodic table it is represented by the symbol S. It is an abundant, multivalent non-metal. Under normal conditions, sulfur atoms form cyclic octatomic molecules with chemical formula S8. Elemental sulfur is a bright yellow...
, and Nickel
Nickel
Nickel is a chemical element with the chemical symbol Ni and atomic number 28. It is a silvery-white lustrous metal with a slight golden tinge. Nickel belongs to the transition metals and is hard and ductile...
.

