
Citrate
Encyclopedia
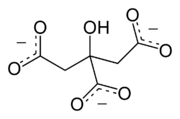
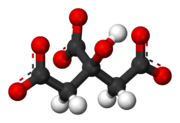
A citrate can refer either to the conjugate base of citric acid
Citric acid
Citric acid is a weak organic acid. It is a natural preservative/conservative and is also used to add an acidic, or sour, taste to foods and soft drinks...
, (C3H5O(COO)33−), or to the ester
Ester
Esters are chemical compounds derived by reacting an oxoacid with a hydroxyl compound such as an alcohol or phenol. Esters are usually derived from an inorganic acid or organic acid in which at least one -OH group is replaced by an -O-alkyl group, and most commonly from carboxylic acids and...
s of citric acid. An example of the former, a salt is trisodium citrate; an ester is triethyl citrate
Triethyl citrate
Triethyl citrate is an ester of citric acid. It is a colorless, odorless liquid used as a food additive to stabilize foams, especially as whipping aid for egg white. It is also used in pharmaceutical coatings and plastics....
.
Other citric acid ions
Since citric acid is a multifunctional acidAcid
An acid is a substance which reacts with a base. Commonly, acids can be identified as tasting sour, reacting with metals such as calcium, and bases like sodium carbonate. Aqueous acids have a pH of less than 7, where an acid of lower pH is typically stronger, and turn blue litmus paper red...
, intermediate ions exist, hydrogen citrate ion, HC6H5O72− and dihydrogen citrate ion, H2C6H5O7−. These may form salts as well, called acid salt
Acid salt
Acid salt is a somewhat obscure term for a class of salts formed by the partial neutralization of diprotic or polyprotic acids. Because the parent acid is only partially neutralized, one or more replaceable hydrogen atoms remain. Typical acid salts have one or more alkali metal ions as well as one...
s.
Acidity
Salts of the hydrogen citrate ions are weakly acidic, while salts of the citrate ion itself (with an inert cation such as sodium ion) are weakly basic.Buffering
As a weak acid, citrate can be used as a component in buffer solutionBuffer solution
A buffer solution is an aqueous solution consisting of a mixture of a weak acid and its conjugate base or a weak base and its conjugate acid. It has the property that the pH of the solution changes very little when a small amount of strong acid or base is added to it. Buffer solutions are used as a...
s, including the commonly used SSC 20X hybridization buffer. This buffer uses sodium citrate and sodium chloride to maintain a neutral 7.0 pH. Other buffers may use a mixture of sodium citrate and citric acid – canonical buffer tables compiled for biochemical studies describe solutions of citrate and acid for buffer pHs of between 3.0 and 6.2.
Chelating
Citric acid can act as a mild chelating agent; citrate, usually in the form of trisodium citrate, may be given as an anticoagulantAnticoagulant
An anticoagulant is a substance that prevents coagulation of blood. A group of pharmaceuticals called anticoagulants can be used in vivo as a medication for thrombotic disorders. Some anticoagulants are used in medical equipment, such as test tubes, blood transfusion bags, and renal dialysis...
, because it chelates calcium ions, and therefore inhibits coagulation
Coagulation
Coagulation is a complex process by which blood forms clots. It is an important part of hemostasis, the cessation of blood loss from a damaged vessel, wherein a damaged blood vessel wall is covered by a platelet and fibrin-containing clot to stop bleeding and begin repair of the damaged vessel...
. Another application is in the form of iron(II) citrate
Iron(II) citrate
Ferrous citrate, or iron citrate, is a chemical compound consisting of one atom of iron and two citrate anions. It has the chemical formula FeC6H6O7....
as a nutritional supplement. Here, the benefit is the solubility as a chelate of the otherwise mostly insoluble iron.
Metabolism
Citrate is an intermediate in the TCA (Krebs) cycle. After the pyruvate dehydrogenase complex forms acetyl-CoAAcetyl-CoA
Acetyl coenzyme A or acetyl-CoA is an important molecule in metabolism, used in many biochemical reactions. Its main function is to convey the carbon atoms within the acetyl group to the citric acid cycle to be oxidized for energy production. In chemical structure, acetyl-CoA is the thioester...
, from pyruvate and five cofactors (thiamine pyrophosphate
Thiamine pyrophosphate
Thiamine pyrophosphate , or thiamine diphosphate , is a thiamine derivative which is produced by the enzyme thiamine pyrophosphatase. Thiamine pyrophosphate is a coenzyme that is present in all living systems, in which it catalyzes several biochemical reactions...
, lipoamide
Lipoamide
Lipoamide is a trivial name for 6,8-dithiooctanoic amide. It is 6,8-dithiooctanoic acid's functional form where the carboxyl group is attached to protein by an amide linkage . Sometimes lipoamide is used to refer to protein bound lipoic acid, but this can be misleading as this is technically...
, FAD
FAD
In biochemistry, flavin adenine dinucleotide is a redox cofactor involved in several important reactions in metabolism. FAD can exist in two different redox states, which it converts between by accepting or donating electrons. The molecule consists of a riboflavin moiety bound to the phosphate...
, NAD
NAD
NAD may refer to:* No abnormality detected, a medical status description* No apparent distress, a status description in childbirth* NAD Electronics, a Canadian audio equipment manufacturer...
+, and CoA
COA
COA can refer to:*Codename Amscray*Cash on Arrival*Cause of action*CedarOpenAccounts*Center of Attention*Certificate of Appealability*Certificate of Approval for marriage or civil partnership in the United Kingdom*Certificate of Authenticity...
), citrate synthase
Citrate synthase
The enzyme citrate synthase exists in nearly all living cells and stands as a pace-making enzyme in the first step of the Citric Acid Cycle . Citrate synthase is localized within eukaryotic cells in the mitochondrial matrix, but is encoded by nuclear DNA rather than mitochondrial...
catalyzes the condensation of oxaloacetate with acetyl CoA to form citrate. Citrate continues in the TCA cycle via aconitase
Aconitase
Aconitase is an enzyme that catalyses the stereo-specific isomerization of citrate to isocitrate via cis-aconitate in the tricarboxylic acid cycle, a non-redox-active process.- Function :...
with the eventual regeneration of oxaloacetate, which can combine with another molecule of acetyl CoA and continue cycling.
Fatty acid synthesis
Citrate can also be transported out of the mitochondria and into the cytoplasm, then broken down into acetyl-CoAAcetyl-CoA
Acetyl coenzyme A or acetyl-CoA is an important molecule in metabolism, used in many biochemical reactions. Its main function is to convey the carbon atoms within the acetyl group to the citric acid cycle to be oxidized for energy production. In chemical structure, acetyl-CoA is the thioester...
for fatty acid synthesis
Fatty acid synthesis
Fatty acid synthesis is the creation of fatty acids from acetyl-CoA and malonyl-CoA precursors through action of enzymes called fatty acid synthases...
. Citrate is a positive modulator of this conversion, and allosterically regulates the enzyme acetyl-CoA carboxylase
Acetyl-CoA carboxylase
Acetyl-CoA carboxylase is a biotin-dependent enzyme that catalyzes the irreversible carboxylation of acetyl-CoA to produce malonyl-CoA through its two catalytic activities, biotin carboxylase and carboxyltransferase...
, which is the regulating enzyme in the conversion of acetyl-CoA into malonyl-CoA
Malonyl-CoA
Malonyl-CoA is a coenzyme A derivative.-Functions:It plays a key role in chain elongation in fatty acid biosynthesis and polyketide biosynthesis....
(the commitment step in fatty acid synthesis). In short, citrate is transported to the cytoplasm, converted to acetyl CoA, which is converted into malonyl CoA by the acetyl CoA carboxylase, which is allosterically modulated by citrate.
See also TCA cycle
Role in glycolysis
High concentrations of cytosolic citrate can inhibit phosphofructokinasePhosphofructokinase
Phosphofructokinase-1 is the most important regulatory enzyme of glycolysis. It is an allosteric enzyme made of 4 subunits and controlled by many activators and inhibitors...
, catalyst of one of the rate-limiting steps of glycolysis
Glycolysis
Glycolysis is the metabolic pathway that converts glucose C6H12O6, into pyruvate, CH3COCOO− + H+...
.

