Stefan-Boltzmann constant
Encyclopedia
The Stefan–Boltzmann constant (also Stefan's constant), a physical constant
denoted by the Greek letter σ
, is the constant of proportionality in the Stefan–Boltzmann law: the total energy
radiated per unit surface area of a black body
in unit time is proportional to the fourth power of the thermodynamic temperature
.
The value of the Stefan–Boltzmann constant is given in SI units
by
In cgs units the Stefan-Boltzmann constant is: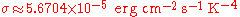 .
.
In US customary
units the Stefan-Boltzmann constant is : .
.
The value of the Stefan–Boltzmann constant is derivable as well as experimentally determinable; see Stefan–Boltzmann law for details. It can be defined in terms of the Boltzmann constant as:
where:
The CODATA recommended value is calculated from the measured value of the gas constant
: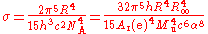
where:
A related constant is the radiation constant (or radiation density constant) a which is given by: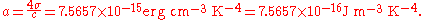
Physical constant
A physical constant is a physical quantity that is generally believed to be both universal in nature and constant in time. It can be contrasted with a mathematical constant, which is a fixed numerical value but does not directly involve any physical measurement.There are many physical constants in...
denoted by the Greek letter σ
Sigma
Sigma is the eighteenth letter of the Greek alphabet, and carries the 'S' sound. In the system of Greek numerals it has a value of 200. When used at the end of a word, and the word is not all upper case, the final form is used, e.g...
, is the constant of proportionality in the Stefan–Boltzmann law: the total energy
Energy
In physics, energy is an indirectly observed quantity. It is often understood as the ability a physical system has to do work on other physical systems...
radiated per unit surface area of a black body
Black body
A black body is an idealized physical body that absorbs all incident electromagnetic radiation. Because of this perfect absorptivity at all wavelengths, a black body is also the best possible emitter of thermal radiation, which it radiates incandescently in a characteristic, continuous spectrum...
in unit time is proportional to the fourth power of the thermodynamic temperature
Thermodynamic temperature
Thermodynamic temperature is the absolute measure of temperature and is one of the principal parameters of thermodynamics. Thermodynamic temperature is an "absolute" scale because it is the measure of the fundamental property underlying temperature: its null or zero point, absolute zero, is the...
.
The value of the Stefan–Boltzmann constant is given in SI units
International System of Units
The International System of Units is the modern form of the metric system and is generally a system of units of measurement devised around seven base units and the convenience of the number ten. The older metric system included several groups of units...
by
In cgs units the Stefan-Boltzmann constant is:
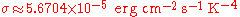 .
.In US customary
United States customary units
United States customary units are a system of measurements commonly used in the United States. Many U.S. units are virtually identical to their imperial counterparts, but the U.S. customary system developed from English units used in the British Empire before the system of imperial units was...
units the Stefan-Boltzmann constant is :
 .
.The value of the Stefan–Boltzmann constant is derivable as well as experimentally determinable; see Stefan–Boltzmann law for details. It can be defined in terms of the Boltzmann constant as:

where:
- kB is the Boltzmann constant;
- h is the Planck constantPlanck constantThe Planck constant , also called Planck's constant, is a physical constant reflecting the sizes of energy quanta in quantum mechanics. It is named after Max Planck, one of the founders of quantum theory, who discovered it in 1899...
; - ħ is the reduced Planck constant;
- c is the speed of lightSpeed of lightThe speed of light in vacuum, usually denoted by c, is a physical constant important in many areas of physics. Its value is 299,792,458 metres per second, a figure that is exact since the length of the metre is defined from this constant and the international standard for time...
in vacuum.
The CODATA recommended value is calculated from the measured value of the gas constant
Gas constant
The gas constant is a physical constant which is featured in many fundamental equations in the physical sciences, such as the ideal gas law and the Nernst equation. It is equivalent to the Boltzmann constant, but expressed in units of energy The gas constant (also known as the molar, universal,...
:
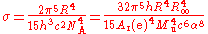
where:
- R is the Universal gas constant;
- NA is the Avogadro constant;
- R∞ is the Rydberg constantRydberg constantThe Rydberg constant, symbol R∞, named after the Swedish physicist Johannes Rydberg, is a physical constant relating to atomic spectra in the science of spectroscopy. Rydberg initially determined its value empirically from spectroscopy, but Niels Bohr later showed that its value could be calculated...
; - Ar(e) is the "relative atomic mass" of the electronElectronThe electron is a subatomic particle with a negative elementary electric charge. It has no known components or substructure; in other words, it is generally thought to be an elementary particle. An electron has a mass that is approximately 1/1836 that of the proton...
; - Mu is the molar mass constantMolar mass constantThe molar mass constant, symbol Mu, is a physical constant which relates atomic weight and molar mass. Its value is defined to be 1 g/mol in SI units....
(1 g/mol by definition); - α is the fine structure constant.
A related constant is the radiation constant (or radiation density constant) a which is given by: