.gif)
Spin states (d electrons)
Encyclopedia
Spin states when describing transition metal
coordination complexes refers to the potential spin configurations of the metal centers d electrons. In many molecules these spin states vary between high-spin and low-spin configurations. These configurations can be understood through the two major models used to describe coordination complexes; ligand field theory
, which is an application of molecular orbital theory
to transition metals, and crystal field theory
, which has roots in VSEPR theory
.
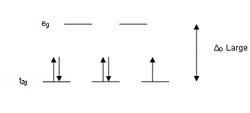 The Δ splitting of the d-orbitals
The Δ splitting of the d-orbitals
plays an important role in the electron spin state of a coordination complex. There are three factors that affect the Δ: the period of the metal center, the charge of the metal center, and the field strength of the complex's ligands as described by the spectrochemical series
.
In order for low spin splitting to occur, the energy cost of placing an electron into an already singly occupied orbital must be less than the cost of placing the additional electron into an eg orbital at an energy cost of Δ. If the energy required to pair two electrons is greater than the energy cost of placing an electron in an eg, Δ, high spin splitting occurs.
If the separation between the orbitals is large, then the lower energy orbitals are completely filled before population of the higher orbitals according to the Aufbau principle
. Complexes such as this are called "low-spin" since filling an orbital matches electrons and reduces the total electron spin. If the separation between the orbitals is small enough then it is easier to put electrons into the higher energy orbitals than it is to put two into the same low-energy orbital, because of the repulsion resulting from matching two electrons in the same orbital. So, one electron is put into each of the five d-orbitals before any pairing occurs in accord with Hund's rule resulting in what is known as a "high-spin" complex. Complexes such as this are called "high-spin" since populating the upper orbital avoids matches between electrons with opposite spin.
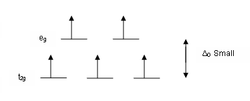 Within a transition metal group moving down the series corresponds with in an increase in Δ. The observed result is larger Δ splitting for complexes in octahedral geometries based around transition metal centers of the second or third row, periods 5 and 6 respectively. This Δ splitting is generally large enough that these complexes do not exist as high-spin state. This is true even when the metal center is coordinated to weak field ligands. It is only octahedral coordination complexes which are centered around first row transition metals that fluctuate between high and low-spin states.
Within a transition metal group moving down the series corresponds with in an increase in Δ. The observed result is larger Δ splitting for complexes in octahedral geometries based around transition metal centers of the second or third row, periods 5 and 6 respectively. This Δ splitting is generally large enough that these complexes do not exist as high-spin state. This is true even when the metal center is coordinated to weak field ligands. It is only octahedral coordination complexes which are centered around first row transition metals that fluctuate between high and low-spin states.
The charge of the metal center plays a role in the ligand field and the Δ splitting. For example, Fe2+ and Co3+ are both d6; however, the higher charge of Co3+ creates a stronger ligand field than Fe2+. All other things being equal, Fe2+ is more likely to be high spin than Co3+.
Ligands also affect the magnitude of Δ splitting of the d-orbitals
according to their field strength as described by the spectrochemical series
. Strong-field ligands, such as CN− and CO, increase the Δ splitting and are more likely to be low-spin. Weak-field ligands, such as I− and Br− cause a smaller Δ splitting and are more likely to be high-spin.
is essentially the same as the crystal field theory
explanation. However the explanation of why the orbitals split is different accordingly with each model and requires translation.
(special version of electron configuration
) with the possibility of holding a high spin or low spin state is octahedral d4 since it has more than the 3 electrons to fill the non bonding d orbitals according to ligand field theory or the stabilized d orbitals according to crystal field splitting. The spin state of the complex also affects an atom's ionic radius
.
d4:Octahedral high-spin: 4 unpaired electrons, paramagnetic, substitutionally labile. Includes Cr2+ ionic radius
80 pm
, Mn3+ ionic radius
64.5 pm
.
d5:Octahedral high-spin: 5 unpaired electrons, paramagnetic, substitutionally labile. Includes Fe3+ ionic radius
64.5 pm
.
d6:Octahedral high-spin: 4 unpaired electrons, paramagnetic, substitutionally labile. Includes Fe2+ ionic radius
78 pm
, Co3+ ionic radius
61 pm
.
d7:Octahedral high-spin: 3 unpaired electrons, paramagnetic, substitutionally labile. Includes Co2+ ionic radius
74.5 pm
, Ni3+ ionic radius
60 pm
.
d8:Octahedral high-spin: 2 unpaired electrons, paramagnetic, substitutionally labile. Includes Ni2+ ionic radius
69 pm
.
Transition metal
The term transition metal has two possible meanings:*The IUPAC definition states that a transition metal is "an element whose atom has an incomplete d sub-shell, or which can give rise to cations with an incomplete d sub-shell." Group 12 elements are not transition metals in this definition.*Some...
coordination complexes refers to the potential spin configurations of the metal centers d electrons. In many molecules these spin states vary between high-spin and low-spin configurations. These configurations can be understood through the two major models used to describe coordination complexes; ligand field theory
Ligand field theory
Ligand field theory describes the bonding, orbital arrangement, and other characteristics of coordination complexes. It represents an application of molecular orbital theory to transition metal complexes. A transition metal ion has nine valence atomic orbitals, five d, one s, and three p orbitals...
, which is an application of molecular orbital theory
Molecular orbital theory
In chemistry, molecular orbital theory is a method for determining molecular structure in which electrons are not assigned to individual bonds between atoms, but are treated as moving under the influence of the nuclei in the whole molecule...
to transition metals, and crystal field theory
Crystal field theory
Crystal field theory is a model that describes the electronic structure of transition metal compounds, all of which can be considered coordination complexes. CFT successfully accounts for some magnetic properties, colours, hydration enthalpies, and spinel structures of transition metal complexes,...
, which has roots in VSEPR theory
VSEPR theory
Valence shell electron pair repulsion theory is a model in chemistry used to predict the shape of individual molecules based upon the extent of electron-pair electrostatic repulsion. It is also named Gillespie–Nyholm theory after its two main developers...
.
Octahedral complexes

Atomic orbital
An atomic orbital is a mathematical function that describes the wave-like behavior of either one electron or a pair of electrons in an atom. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus...
plays an important role in the electron spin state of a coordination complex. There are three factors that affect the Δ: the period of the metal center, the charge of the metal center, and the field strength of the complex's ligands as described by the spectrochemical series
Spectrochemical series
A spectrochemical series is a list of ligands ordered on ligand strength and a list of metal ions based on oxidation number, group and its identity...
.
In order for low spin splitting to occur, the energy cost of placing an electron into an already singly occupied orbital must be less than the cost of placing the additional electron into an eg orbital at an energy cost of Δ. If the energy required to pair two electrons is greater than the energy cost of placing an electron in an eg, Δ, high spin splitting occurs.
If the separation between the orbitals is large, then the lower energy orbitals are completely filled before population of the higher orbitals according to the Aufbau principle
Aufbau principle
The Aufbau principle is used to determine the electron configuration of an atom, molecule or ion. The principle postulates a hypothetical process in which an atom is "built up" by progressively adding electrons...
. Complexes such as this are called "low-spin" since filling an orbital matches electrons and reduces the total electron spin. If the separation between the orbitals is small enough then it is easier to put electrons into the higher energy orbitals than it is to put two into the same low-energy orbital, because of the repulsion resulting from matching two electrons in the same orbital. So, one electron is put into each of the five d-orbitals before any pairing occurs in accord with Hund's rule resulting in what is known as a "high-spin" complex. Complexes such as this are called "high-spin" since populating the upper orbital avoids matches between electrons with opposite spin.

The charge of the metal center plays a role in the ligand field and the Δ splitting. For example, Fe2+ and Co3+ are both d6; however, the higher charge of Co3+ creates a stronger ligand field than Fe2+. All other things being equal, Fe2+ is more likely to be high spin than Co3+.
Ligands also affect the magnitude of Δ splitting of the d-orbitals
Atomic orbital
An atomic orbital is a mathematical function that describes the wave-like behavior of either one electron or a pair of electrons in an atom. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus...
according to their field strength as described by the spectrochemical series
Spectrochemical series
A spectrochemical series is a list of ligands ordered on ligand strength and a list of metal ions based on oxidation number, group and its identity...
. Strong-field ligands, such as CN− and CO, increase the Δ splitting and are more likely to be low-spin. Weak-field ligands, such as I− and Br− cause a smaller Δ splitting and are more likely to be high-spin.
Tetrahedral complexes
The Δ splitting energy for tetrahedral metal complexes (four ligands), Δtet is smaller than that for an octahedral complex. Therefore, it is rare to have a Δtet large enough to cause electrons to pair before filling high orbitals. Thus, tetrahedral complexes are usually high-spin. "There are no known ligands powerful enough to produce the strong-field case in a tetrahedral complex" (Transition metals and Coordination Chemistry:The Crystal field Model by Steven S. Zumdahl. Chemical Principles)Square planar complexes
Most spin state transitions are between the same geometry, namely octahedral. However, in the case of d8 complexes is a shift in geometry between spin states. There is no possible difference between the high and low-spin states in the d8 octahedral complexes, however d8 complexes are able to shift from paramagnetic tetrahedral geometry to a diamagnetic low-spin square planar geometry.Ligand field theory vs Crystal field theory
The rationale for why the spin states exist according to ligand field theoryLigand field theory
Ligand field theory describes the bonding, orbital arrangement, and other characteristics of coordination complexes. It represents an application of molecular orbital theory to transition metal complexes. A transition metal ion has nine valence atomic orbitals, five d, one s, and three p orbitals...
is essentially the same as the crystal field theory
Crystal field theory
Crystal field theory is a model that describes the electronic structure of transition metal compounds, all of which can be considered coordination complexes. CFT successfully accounts for some magnetic properties, colours, hydration enthalpies, and spinel structures of transition metal complexes,...
explanation. However the explanation of why the orbitals split is different accordingly with each model and requires translation.
High-spin and low-spin systems
The first d electron countD electron count
The d electron count is a chemistry formalism used to describe the electron configuration of the valence electrons of a transition metal center in a coordination complex. The d electron count is an effective way to understand the geometry and reactivity of transition metal complexes...
(special version of electron configuration
Electron configuration
In atomic physics and quantum chemistry, electron configuration is the arrangement of electrons of an atom, a molecule, or other physical structure...
) with the possibility of holding a high spin or low spin state is octahedral d4 since it has more than the 3 electrons to fill the non bonding d orbitals according to ligand field theory or the stabilized d orbitals according to crystal field splitting. The spin state of the complex also affects an atom's ionic radius
Ionic radius
Ionic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
.
d4:Octahedral high-spin: 4 unpaired electrons, paramagnetic, substitutionally labile. Includes Cr2+ ionic radius
Ionic radius
Ionic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
80 pm
Picometre
A picometre is a unit of length in the metric system, equal to one trillionth, i.e. of a metre, which is the current SI base unit of length...
, Mn3+ ionic radius
Ionic radius
Ionic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
64.5 pm
Picometre
A picometre is a unit of length in the metric system, equal to one trillionth, i.e. of a metre, which is the current SI base unit of length...
.
- Octahedral low-spin: 2 unpaired electrons, paramagnetic, substitutionally inert. Includes Cr2+ ionic radiusIonic radiusIonic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
73 pmPicometreA picometre is a unit of length in the metric system, equal to one trillionth, i.e. of a metre, which is the current SI base unit of length...
, Mn3+ ionic radiusIonic radiusIonic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
58 pmPicometreA picometre is a unit of length in the metric system, equal to one trillionth, i.e. of a metre, which is the current SI base unit of length...
.
d5:Octahedral high-spin: 5 unpaired electrons, paramagnetic, substitutionally labile. Includes Fe3+ ionic radius
Ionic radius
Ionic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
64.5 pm
Picometre
A picometre is a unit of length in the metric system, equal to one trillionth, i.e. of a metre, which is the current SI base unit of length...
.
- Octahedral low-spin: 1 unpaired electron, paramagnetic, substitutionally inert. Includes Fe3+ ionic radiusIonic radiusIonic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
55 pmPicometreA picometre is a unit of length in the metric system, equal to one trillionth, i.e. of a metre, which is the current SI base unit of length...
.
d6:Octahedral high-spin: 4 unpaired electrons, paramagnetic, substitutionally labile. Includes Fe2+ ionic radius
Ionic radius
Ionic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
78 pm
Picometre
A picometre is a unit of length in the metric system, equal to one trillionth, i.e. of a metre, which is the current SI base unit of length...
, Co3+ ionic radius
Ionic radius
Ionic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
61 pm
Picometre
A picometre is a unit of length in the metric system, equal to one trillionth, i.e. of a metre, which is the current SI base unit of length...
.
- Octahedral low-spin: no unpaired electrons, diamagnetic, substitutionally inert. Includes Fe2+ ionic radiusIonic radiusIonic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
62 pmPicometreA picometre is a unit of length in the metric system, equal to one trillionth, i.e. of a metre, which is the current SI base unit of length...
, Co3+ ionic radiusIonic radiusIonic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
54.5 pmPicometreA picometre is a unit of length in the metric system, equal to one trillionth, i.e. of a metre, which is the current SI base unit of length...
, Ni4+ ionic radiusIonic radiusIonic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
48 pmPicometreA picometre is a unit of length in the metric system, equal to one trillionth, i.e. of a metre, which is the current SI base unit of length...
.
d7:Octahedral high-spin: 3 unpaired electrons, paramagnetic, substitutionally labile. Includes Co2+ ionic radius
Ionic radius
Ionic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
74.5 pm
Picometre
A picometre is a unit of length in the metric system, equal to one trillionth, i.e. of a metre, which is the current SI base unit of length...
, Ni3+ ionic radius
Ionic radius
Ionic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
60 pm
Picometre
A picometre is a unit of length in the metric system, equal to one trillionth, i.e. of a metre, which is the current SI base unit of length...
.
- Octahedral low-spin:1 unpaired electron, paramagnetic, substitutionally labile. Includes Co2+ ionic radiusIonic radiusIonic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
65 pmPicometreA picometre is a unit of length in the metric system, equal to one trillionth, i.e. of a metre, which is the current SI base unit of length...
, Ni3+ ionic radiusIonic radiusIonic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
56 pmPicometreA picometre is a unit of length in the metric system, equal to one trillionth, i.e. of a metre, which is the current SI base unit of length...
.
d8:Octahedral high-spin: 2 unpaired electrons, paramagnetic, substitutionally labile. Includes Ni2+ ionic radius
Ionic radius
Ionic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
69 pm
Picometre
A picometre is a unit of length in the metric system, equal to one trillionth, i.e. of a metre, which is the current SI base unit of length...
.
- Square planar low-spin: no unpaired electrons, diamagnetic, substitutionally inert. Includes Ni2+ ionic radiusIonic radiusIonic radius, rion, is the radius of an atom's ion. Although neither atoms nor ions have sharp boundaries, it is important to treat them as if they are hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice...
49 pmPicometreA picometre is a unit of length in the metric system, equal to one trillionth, i.e. of a metre, which is the current SI base unit of length...
.

