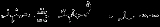
Krapcho decarboxylation
Encyclopedia
The Krapcho decarboxylation is the chemical reaction
of ester
s with halide
anions. The ester must contain an electron-withdrawing group in the beta position, such as β-ketoesters, malonic esters, α-cyanoesters, or α-sulfonylesters. It works best with methyl esters, since it is basically a SN2-reaction at carbon. It is driven by the entropy of the overall reaction, as the byproducts chloromethane
and CO2
are lost as gases. The reaction is a useful synthetic procedure to hydrolyze and decarboxylate malonic esters
which only cleaves one of the ester groups. The alternative way (e.g. basic hydrolysis) destroys both of the ester groups and a subsequent reaction is usually used to regenerate the alkylated ester.
Chemical reaction
A chemical reaction is a process that leads to the transformation of one set of chemical substances to another. Chemical reactions can be either spontaneous, requiring no input of energy, or non-spontaneous, typically following the input of some type of energy, such as heat, light or electricity...
of ester
Ester
Esters are chemical compounds derived by reacting an oxoacid with a hydroxyl compound such as an alcohol or phenol. Esters are usually derived from an inorganic acid or organic acid in which at least one -OH group is replaced by an -O-alkyl group, and most commonly from carboxylic acids and...
s with halide
Halide
A halide is a binary compound, of which one part is a halogen atom and the other part is an element or radical that is less electronegative than the halogen, to make a fluoride, chloride, bromide, iodide, or astatide compound. Many salts are halides...
anions. The ester must contain an electron-withdrawing group in the beta position, such as β-ketoesters, malonic esters, α-cyanoesters, or α-sulfonylesters. It works best with methyl esters, since it is basically a SN2-reaction at carbon. It is driven by the entropy of the overall reaction, as the byproducts chloromethane
Chloromethane
Chloromethane, also called methyl chloride, R-40 or HCC 40, is a chemical compound of the group of organic compounds called haloalkanes. It was once widely used as a refrigerant. It is a colorless extremely flammable gas with a minorly sweet odor, which is, however, detected at possibly toxic levels...
and CO2
Carbon dioxide
Carbon dioxide is a naturally occurring chemical compound composed of two oxygen atoms covalently bonded to a single carbon atom...
are lost as gases. The reaction is a useful synthetic procedure to hydrolyze and decarboxylate malonic esters
Malonic ester synthesis
The malonic ester synthesis is a chemical reaction where diethyl malonate or another ester of malonic acid is alkylated at the carbon alpha to both carbonyl groups, and then converted to a substituted acetic acid. The major drawback of malonic ester synthesis is that the alkylation stage can also...
which only cleaves one of the ester groups. The alternative way (e.g. basic hydrolysis) destroys both of the ester groups and a subsequent reaction is usually used to regenerate the alkylated ester.

