Sulfonic acid
Overview
Organosulfur compounds
Organosulfur compounds are organic compounds that contain sulfur. They are often associated with foul odours, but many of the sweetest compounds known are organosulfur derivatives. Nature abounds with organosulfur compounds—sulfur is essential for life. Two of the 20 common amino acids are...
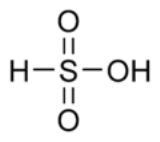
with the general formula RS(=O)2–OH, where R is an alkyl or aryl
Aryl
In the context of organic molecules, aryl refers to any functional group or substituent derived from an aromatic ring, be it phenyl, naphthyl, thienyl, indolyl, etc....
. The formal part of acid, HS(=O)2–OH, are formally derivatives of the "parent" inorganic compound
Inorganic compound
Inorganic compounds have traditionally been considered to be of inanimate, non-biological origin. In contrast, organic compounds have an explicit biological origin. However, over the past century, the classification of inorganic vs organic compounds has become less important to scientists,...
with the formula HSO2(OH).
Sulfonic acid is produced by the process of sulfonation. Usually the sulfonating agent is sulfur trioxide
Sulfur trioxide
Sulfur trioxide is the chemical compound with the formula SO3. In the gaseous form, this species is a significant pollutant, being the primary agent in acid rain. It is prepared on massive scales as a precursor to sulfuric acid.-Structure and bonding:Gaseous SO3 is a trigonal planar molecule of...
.
Unanswered Questions

