Diazene
Encyclopedia
Diazene, also called diimine or diimide, is a compound having the formula (NH)2. It exists as two geometric isomers, E and Z. Diazene is also the parent member of the entire class of azo compound
s with the formula (NR)2, where R is an organyl group. Thus, azobenzene
is an example of an organic diazene.
with hydrogen peroxide or air. Alternatively the decarboxylation of azodicarboxylic acid affords diimide:2 → (NH)2 + 2 CO2
Diazene can also be efficiently generated by elimination of sulfonohydrazides using a suitable base. For example 2,4,6-triisopropylbenzenesulfonohydrazide eliminates diazene upon treatment with sodium bicarbonate
, a very mild base.
Because of its instability, diimide is generated and used in-situ. A mixture of both the cis (Z-) and trans (E-) isomers is produced, both of which are unstable. The isomerisation can be slow. The trans - cis equilibrium is driven to the right due to the rapid consumption of the cis isomer upon reaction with the unsaturated substrate. Some procedures call for the addition of carboxylic acids, which catalyse the cis-trans isomerization. Diimide decomposes readily via disproportionation:
Because of this competing decomposition reaction, reductions with diimide typically require a large excess of the precursor reagent.
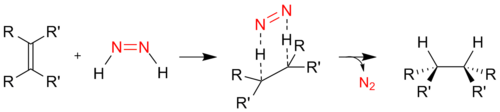
. It hydrogenates alkenes and alkynes with selective delivery of hydrogen from one face of the substrate resulting in the same stereoselectivty as metal-catalysed syn addition of H2. The only coproduct released is nitrogen gas. Although the method is cumbersome, the use of diimide avoids the need for high pressures or potentially explosive hydrogen gas and metal catalysts, which can be expensive. The hydrogenation mechanism involves a six-membered C2H2N2 transition state:
s that would interfere with normal catalytic hydrogenation. Thus, peroxide
s, alkyl halides, and thiol
s are tolerated by diimide, but these same groups would typically be degraded by metal catalysts. The reagent preferentially reduces alkynes and unhindered or strained alkenes to the corresponding alkenes and alkanes.
Azo compound
Azo compounds are compounds bearing the functional group R-N=N-R', in which R and R' can be either aryl or alkyl. IUPAC defines azo compounds as: "Derivatives of diazene , HN=NH, wherein both hydrogens are substituted by hydrocarbyl groups, e.g. PhN=NPh azobenzene or diphenyldiazene." The more...
s with the formula (NR)2, where R is an organyl group. Thus, azobenzene
Azobenzene
Azobenzene is a chemical compound composed of two phenyl rings linked by a N=N double bond. It is the best known example of an azo compound. The term 'azobenzene' or simply 'azo' is often used to refer to a wide class of molecules that share the core azobenzene structure, with different chemical...
is an example of an organic diazene.
Synthesis
The traditional route to diimide involves oxidation of hydrazineHydrazine
Hydrazine is an inorganic compound with the formula N2H4. It is a colourless flammable liquid with an ammonia-like odor. Hydrazine is highly toxic and dangerously unstable unless handled in solution. Approximately 260,000 tons are manufactured annually...
with hydrogen peroxide or air. Alternatively the decarboxylation of azodicarboxylic acid affords diimide:2 → (NH)2 + 2 CO2
Diazene can also be efficiently generated by elimination of sulfonohydrazides using a suitable base. For example 2,4,6-triisopropylbenzenesulfonohydrazide eliminates diazene upon treatment with sodium bicarbonate
Sodium bicarbonate
Sodium bicarbonate or sodium hydrogen carbonate is the chemical compound with the formula Na HCO3. Sodium bicarbonate is a white solid that is crystalline but often appears as a fine powder. It has a slightly salty, alkaline taste resembling that of washing soda . The natural mineral form is...
, a very mild base.
Because of its instability, diimide is generated and used in-situ. A mixture of both the cis (Z-) and trans (E-) isomers is produced, both of which are unstable. The isomerisation can be slow. The trans - cis equilibrium is driven to the right due to the rapid consumption of the cis isomer upon reaction with the unsaturated substrate. Some procedures call for the addition of carboxylic acids, which catalyse the cis-trans isomerization. Diimide decomposes readily via disproportionation:
- 2 (NH)2 → (NH2)2 + N2
Because of this competing decomposition reaction, reductions with diimide typically require a large excess of the precursor reagent.
Applications to organic synthesis
cis-Diazene is occasionally useful as a reagent in organic synthesisOrganic synthesis
Organic synthesis is a special branch of chemical synthesis and is concerned with the construction of organic compounds via organic reactions. Organic molecules can often contain a higher level of complexity compared to purely inorganic compounds, so the synthesis of organic compounds has...
. It hydrogenates alkenes and alkynes with selective delivery of hydrogen from one face of the substrate resulting in the same stereoselectivty as metal-catalysed syn addition of H2. The only coproduct released is nitrogen gas. Although the method is cumbersome, the use of diimide avoids the need for high pressures or potentially explosive hydrogen gas and metal catalysts, which can be expensive. The hydrogenation mechanism involves a six-membered C2H2N2 transition state:
Selectivity
Diazene is advantageous because of it selectively reduces alkenes and alkynes and is nonreactive toward many functional groupFunctional group
In organic chemistry, functional groups are specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules. The same functional group will undergo the same or similar chemical reaction regardless of the size of the molecule it is a part of...
s that would interfere with normal catalytic hydrogenation. Thus, peroxide
Peroxide
A peroxide is a compound containing an oxygen–oxygen single bond or the peroxide anion .The O−O group is called the peroxide group or peroxo group. In contrast to oxide ions, the oxygen atoms in the peroxide ion have an oxidation state of −1.The simplest stable peroxide is hydrogen peroxide...
s, alkyl halides, and thiol
Thiol
In organic chemistry, a thiol is an organosulfur compound that contains a carbon-bonded sulfhydryl group...
s are tolerated by diimide, but these same groups would typically be degraded by metal catalysts. The reagent preferentially reduces alkynes and unhindered or strained alkenes to the corresponding alkenes and alkanes.