Asymmetric carbon
Encyclopedia
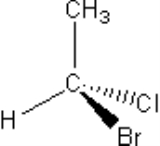
An asymmetric carbon atom is a carbon
atom
that is attached to four different types of atom or four different groups of atoms. Knowing the number of asymmetric carbon atoms, one can calculate the maximum possible number of stereoisomers for any given molecule as follows:
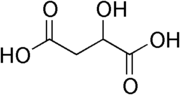
As an example, malic acid
has 4 carbon atoms but just one of them is asymmetric:
A tetrose
with 2 asymmetric carbon atoms has 22 = 4 stereoisomers:
An aldopentose with 3 asymmetric carbon atoms has 23 = 8 stereoisomers:
An aldohexose
with 4 asymmetric carbon atoms has 24 = 16 stereoisomers:
The four groups of atoms attached to the carbon atom can be arranged in space in two different ways that are mirror images of each other, and which lead to so-called left-handed and right-handed versions of the same molecule. Molecules that cannot be superimposed on their own mirror image are said to be chiral
.
Carbon
Carbon is the chemical element with symbol C and atomic number 6. As a member of group 14 on the periodic table, it is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds...
atom
Atom
The atom is a basic unit of matter that consists of a dense central nucleus surrounded by a cloud of negatively charged electrons. The atomic nucleus contains a mix of positively charged protons and electrically neutral neutrons...
that is attached to four different types of atom or four different groups of atoms. Knowing the number of asymmetric carbon atoms, one can calculate the maximum possible number of stereoisomers for any given molecule as follows:
- If n is the number of asymmetric carbon atoms then the maximum number of isomers = 2n
As an example, malic acid
Malic acid
Malic acid is an organic compound with the formula HO2CCH2CHOHCO2H. It is a dicarboxylic acid which is made by all living organisms, contributes to the pleasantly sour taste of fruits, and is used as a food additive. Malic acid has two stereoisomeric forms , though only the L-isomer exists...
has 4 carbon atoms but just one of them is asymmetric:
A tetrose
Tetrose
A tetrose is a monosaccharide with 4 carbon atoms. They have either an aldehyde functional group in position 1 or a ketone functional group in position 2 ....
with 2 asymmetric carbon atoms has 22 = 4 stereoisomers:
An aldopentose with 3 asymmetric carbon atoms has 23 = 8 stereoisomers:
An aldohexose
Aldohexose
An aldohexose is a hexose with an aldehyde group on one end.The aldohexoses have four chiral centres for a total of 16 possible aldohexose stereoisomers . Of these, only three commonly occur in nature: D-glucose, D-galactose, and D-mannose...
with 4 asymmetric carbon atoms has 24 = 16 stereoisomers:
The four groups of atoms attached to the carbon atom can be arranged in space in two different ways that are mirror images of each other, and which lead to so-called left-handed and right-handed versions of the same molecule. Molecules that cannot be superimposed on their own mirror image are said to be chiral
Chirality (chemistry)
A chiral molecule is a type of molecule that lacks an internal plane of symmetry and thus has a non-superimposable mirror image. The feature that is most often the cause of chirality in molecules is the presence of an asymmetric carbon atom....
.