1novicethinker
I am truly a novice thinker who has been bothered by the seemingly unstable atomic model of electrons orbiting around the various nucleii of all elemental mass. To me this is illogical since the atomic models of all natural occurring elements look unstable with respect to the symmetry of these models. For example, I can't see how the electrons spinning around nucleii could even allow for isotopic nucleii transformatioms where the nucleus count changes! I have come to pose the interesting question above in the subject title.
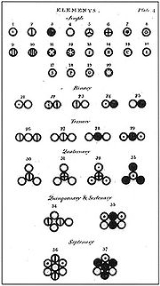
I can visualize any nucleus of any element or any associated isotope, as having within it, symmetrical hollow conical electromagnetic fields that emanate outward from the centerpoint of the nucleus to the outside.These portholes would exist symmetrically around a field axis or more than one field axis, in the case of heavier elements, that could contain all the oribiting electrons associated with any particular element or isotope of that element. They could exist in levels that are consistent with current atomic theory! In other words, the electrons would exist rotating in "rings" inside the nucleus itself, arranged within these various conical shaped electromagnetic fields that allow electrons open passage to and from the inside of any nucleii! Heavier elements could contain three or more field axes passing symmetrically through the nucleii, each having two conical shaped electromagnetic fields associated with each axis! This means there could be six symmetrical conical electromagnetic fields occurring around 3 separate symmetrical field axes all containing inside orbiting electrons! In this way, each conical field would allow electron passage to and from the nucleii! This may more readily explain continued symmetry for even the most complicated electron ring pattern arrangements. Also, electrons can change energy levels with this atomic model! Respectfully Submitted, 1 novice thinker
I can visualize any nucleus of any element or any associated isotope, as having within it, symmetrical hollow conical electromagnetic fields that emanate outward from the centerpoint of the nucleus to the outside.These portholes would exist symmetrically around a field axis or more than one field axis, in the case of heavier elements, that could contain all the oribiting electrons associated with any particular element or isotope of that element. They could exist in levels that are consistent with current atomic theory! In other words, the electrons would exist rotating in "rings" inside the nucleus itself, arranged within these various conical shaped electromagnetic fields that allow electrons open passage to and from the inside of any nucleii! Heavier elements could contain three or more field axes passing symmetrically through the nucleii, each having two conical shaped electromagnetic fields associated with each axis! This means there could be six symmetrical conical electromagnetic fields occurring around 3 separate symmetrical field axes all containing inside orbiting electrons! In this way, each conical field would allow electron passage to and from the nucleii! This may more readily explain continued symmetry for even the most complicated electron ring pattern arrangements. Also, electrons can change energy levels with this atomic model! Respectfully Submitted, 1 novice thinker